The 3D BioFabrication Facility (BFF) bioprinter from nScrypt, a Florida-based 3D printing system manufacturer, and spaceflight equipment developer Techshot is set to launch on the International Space Station (ISS) next month.
Said to be the first 3D printer capable of manufacturing human tissue in microgravity conditions, the BFF will be launched aboard the SpaceX CRS-18 cargo mission from Florida’s Cape Canaveral Air Force Station. The partners hope that 3D bioprinting in space will be capable of producing self-supporting tissues that could lead to the development of therapeutic treatments.
“The concept of developing tissue or an organ in space with a 3D bioprinter has been with us for years,” said John Vellinger, President and CEO of Techshot.
“From the technological and biological perspectives, making it real has been a painstaking process of experiments and testing. To see this coming together is an amazing thing for the team, as well as for all of medical science.”
Extraterrestrial 3D bioprinting
Based in Orlando, Florida, nScrypt is spin out of Sciperio Inc which, under a DARPA contract, developed an award-winning bioprinter in 2003. Techshot, on the other hand, is located in Greenville, Indiana, and has almost three decades of experience in the development of spaceflight equipment.
Vellinger’s first project to earn its place on a NASA mission was a KFC-sponsored payload intended to study how microgravity in space affects the growth of chicken embryos. In 2016, with help from bio-ink startup, Bioficial Organs, the companies were able to successfully 3D print cardiac and vascular structures in zero gravity.
Last year, the following phase of a NASA sponsored mission to 3D print human organs and tissues in space was announced to be launched in February 2019. Now, the quest of manufacturing hearts and other organs via a 3D bioprinter is continuing in July. This ultimately aims to reduce the current shortage of donor organs worldwide.

Space-made cell structures
During the companies years of research, it was found that 3D bioprinted soft, easily flowing biomaterials (i.e, human tissue) collapse under their own weight. However, these same materials are able to maintain their shapes when produced in the microgravity environment of space.
Thus, these structures, which will consist of blood vessels and muscle, will be 3D bioprinted in space and placed in a cell-culturing system that strengthens them over time. This will permit them to become viable, self-supporting tissues that will remain solid once back on Earth.
The initial phase for BFF, which could last an estimated two years, will involve creating test prints of cardiac-like tissue of increasing thickness. The following phase will involve an Earth-based evaluation of heart patches manufactured in space under a microscope and potentially in small animals such as rats. This is expected to last through 2024.
Find the latest additive manufacturing news by subscribing to our 3D Printing Industry newsletter and following us Facebook and Twitter.
Also, visit our 3D Printing Jobs board to find out more about opportunities in additive manufacturing.
Featured image shows the 3D BioFabrication Facility (BFF) bioprinter. Photo via Techshot.
HP, manufacturers of the award-winning Multi Jet Fusion (MJF) 3D printers, and SmileDirectClub, a Nashville-based teledentistry company, have announced a partnership to increase the production of its customized clear aligners at RAPID + TCT.
“SmileDirectClub is digitally transforming the traditional orthodontics industry, making it more personal, affordable, and convenient for millions of consumers to achieve a smile they’ll love,” said Alex Fenkell, co-founder of SmileDirectClub.
“HP’s breakthrough 3D printing and data intelligence platform makes this level of disruption possible for us, pushing productivity, quality, and manufacturing predictability to unprecedented levels, all with economics that allows us to pass on savings to the consumers seeking treatment using our teledentistry platform.”

Disrupting the dental sector
Founded in 2014, SmileDirectClub creates personalized clear aligners in collaboration with its digital network of state-licensed dentists and orthodontists. The company has reportedly served over 500,000 customers in the U.S., Canada, and Puerto Rico, and intends to expand to Australia and the UK this year. In comparison a competitor, California-based Align Technology is offering Invisalign to over 45 countries where it is treating more than 2.5 million patients.
According to SmileDirectClub, approximately 80% of people in the U.S. could benefit from orthodontic care, yet only 1% receive it each year. This lack of access has been attributed to the cost of dental care and absence of orthodontists for certain treatments.
Thus, the company has partnered with HP to expand the reach of inexpensive, personalized clear aligners and other teeth-straightening treatments. Christoph Schell, President of 3D Printing and Digital Manufacturing at HP Inc, added:
“SmileDirectClub and HP are reinventing the future of orthodontics, pushing the boundaries of customized 3D mass production and democratizing access to affordable, high-quality teeth straightening for millions of people.”
“Through this collaboration, HP is helping SmileDirectClub accelerate its growth, enabling a new era of personalized consumer experiences only made possible by industrial 3D printing and digital manufacturing.”

50,000 3D printed aligner molds a day
As a result of this partnership, SmileDirectClub has more than doubled its investment in HP 3D printing solutions. As such, the company will deploy 49 HP Jet Fusion 3D printing systems running 24/7, to produce more than 50,000 unique mouth molds a day, an estimated 20 million molds over the next 12 months. In contrast, according to Joseph Hogan, CEO of Align Technology, over 500,000 unique 3D printed parts for Invisalign clear aligners are produced per day using 3D Systems technology.
Furthermore, HP and SmileDirectClub have announced a new recycling program, where excess 3D material and existing processed plastic mouth molds are recycled and converted into pellets for traditional injection molding. This collaboration has made SmileDirectClub’s production facility the largest HP Multi Jet Fusion 3D Production Factory in the US.
Don’t forget to vote for the 2019 3D Printing Industry Awards.
Also, subscribe to our 3D Printing Industry newsletter and follow us Facebook and Twitter for full coverage of RAPID + TCT. 3D Printing Industry is the only publication reporting all of the news from the event.
Visit our 3D Printing Jobs board to find out more about opportunities in additive manufacturing.
Featured image shows personalized clear aligners. Photo via SmileDirectClub.
Pandorum Technologies Pvt., a Bangalore-based biotechnology company, has used 3D bioprinted cornea tissue to promote scarless healing of wounds in the eye.
“In India alone, there are over a million people suffering from a bilateral loss of vision due to corneal disorders, and at least a few folds more from unilateral corneal blindness,” said Dr. Tuhin Bhowmick, co-founder of Pandorum with a Ph.D. from the Indian Institute of Science.
“We are working to close this gap using a bio-engineering approach through stage-wise development of a platform, which is ultimately aimed to liberate us from the dependencies on human donor cornea,” added Dr. Bhowmick, who is also an alumnus of the European Molecular Biology Labs (EMBL).

Liquid Cornea
Founded in 2011, Pandorum Technologies Pvt. specializes in tissue engineering and regenerative medicine for improved healthcare. In collaboration with India’s LV Prasad Eye Institute, the company created a bio-inspired corneal ink dubbed as ‘Liquid Cornea’ to 3D bioprint corneal lenticule – a disc-shaped piece of corneal tissue which can also be manufactured with synthetic materials.
This is implanted into or on top of the human eye to treat visual impairment due to corneal defect. “Corneal disorders are one of the major causes of blindness worldwide,” continued Dr. Bhowmick.
“Though surgically replacing the opaque tissue with a clear corneal allograft is usually effective in improving vision, there is an acute shortage of cadaveric human corneas available for transplantation. Being able to bio-engineer critical tissues such as the human cornea is a significant milestone.”
Currently, pre-clinical studies involving corneal wound models in rabbits are underway and the team is preparing for pilot human studies in 2020.

A 3D printed eye for an eye
Elsewhere, 3D bioprinting has been used to blindness caused by corneal disease. Recently, scientists at Newcastle University (NCL) have successfully 3D printed gelatinous and multicellular human corneas.
Also, the University of Sydney’s Save Sight Institute and the University of Wollongong (UOW) have developed a 3D corneal biopen which acts as a handheld co-axial 3D printer, extruding bioink directly onto an eye to aid in the regeneration of cells on corneal ulcers. This pen also creates a biological barrier towards ongoing cornea damage caused by infections.
Vote for the 2019 3D Printing Industry Awards.
Subscribe to our 3D printing newsletter and follow us Facebook and Twitter for the latest additive manufacturing updates.
Visit our 3D Printing Jobs board to find out more about opportunities in additive manufacturing.
Featured image shows a 3D Bioprinted Cornea Stromal Lenticule. Photo via Pandorum Technologies Pvt.
Nexxt Spine, an Indiana-based medical device manufacturer, has expanded its metal 3D printing capabilities with the installation of two Concept Laser Mlab 100R systems from GE Additive.
The pair of machines are the fourth and fifth additive manufacturing acquisition from the company since 2017 and will support the design and production of Nexxt Spine’s spinal fusion implants.
“Additive is booming,” stated Alaedeen Abu-Mulaweh, director of engineering at Nexxt Spine.
“We used the first Mlab primarily for R&D purposes, but soon realized that further investment in additive technology could add value not only to our overall growth strategy but also at a clinical application level with the ability to develop implants with very intricate micro-geometries that could maximize healing.”
3D printed spinal implants
Founded in 2009, Nexxt Spine manufactures spinal implants and surgical instruments to innovate and improve upon spinal procedures. With its shift into industrial 3D printing two years ago the company launched the MATRIXX family of porous titanium spinal fusion implants with novel 3D printed cellular scaffolding.
Presently, the company has 16 product lines available within the U.S. and all of its devices are created at its facility in Noblesville. According to the company, the investment in Concept Laser Mlab machines allows its team to control the entire design, production, and distribution process in-house.
“Over the past two years, we have made a seamless jump from R&D to serial production and in doing so have significantly accelerated the time from concept to commercialization,” added Abu-Mulaweh.

Improving cellular healing
Abu-Mulaweh continued, “Titanium – porous or otherwise – is physically incapable of biological remodeling, so using additive to directly mimic the structural randomness of bone doesn’t make a whole lot of sense.”
“Rather than simply looking like bone, Nexxt MATRIXX was designed with functionality in mind to fulfill our vision of actively facilitating the body’s natural power of cellular healing.”
Earlier this year, Nexxt Spine began developing its 3D printed bone healing spinal implants with MTS Systems Corporation, a global manufacturer and supplier of simulations and testing systems.
“We are seeing ongoing adoption of additive manufacturing in the orthopaedic industry and an exciting shift from research and development to serial production,” said Stephan Zeidler, senior global and key accounts director for the medical sector at GE Additive.
“Early innovators like Nexxt Spine are scaling up and there is a significant increase in production volumes. [Its] another great example that shows the power of our Mlab machine.”
Vote for the best “Medical, dental or healthcare application” for the 2019 3D Printing Industry Awards.
Subscribe to our 3D printing newsletter and follow us Facebook and Twitter for the latest additive manufacturing updates.
Visit our 3D Printing Jobs board to find out more about opportunities in additive manufacturing.
Featured image show Nexxt Matrixx Vertebral Body Replacement implant 3D printed in titanium. Image via Nexxt Spine.
3DP4ME, a Jordanian non-profit organization using 3D printing to meet human needs, and World Wide Hearing (WWH) are seeking to raise $200,000 to provide 3D printed hearing-aid molds in the Middle East.
The partner’s previous goal of $100,000 has been increased to develop and manufacture thousands of hearing aid molds a year which will potentially be available worldwide. This new fundraiser has currently accumulated $50,567 in donations.
The Hearing Express Project
Hearing-aids must be tailored to its user to prevent sound leakage and provide maximum comfort. Customizable 3D printed hearing-aids have demonstrated the ability to improve conventional devices to isolate speech and reduce background noise.
Using a portable Lantos 3D Ear Scanner, 3DP4ME and the WWH aim to visit clients in their homes, villages, and refugee camps to collect data for a personalized 3D printed hearing aid ear mold.
“Once we complete a 3D scan of our client, a 3D model is created in real-time on our internet cloud. Meanwhile, back in the lab, our 3D modeler can immediately start preparing the STL files for 3D printing,” stated Andrew De Carpentier, Head of the Hearing Express Project at 3DP4ME.
The molds are produced on the ASIGA PRO 2 3D Printer using SLA. This system is capable of manufacturing up to 35 ear molds in one hour, a significant improvement from making 3 or 4 ear molds by hand in a day.
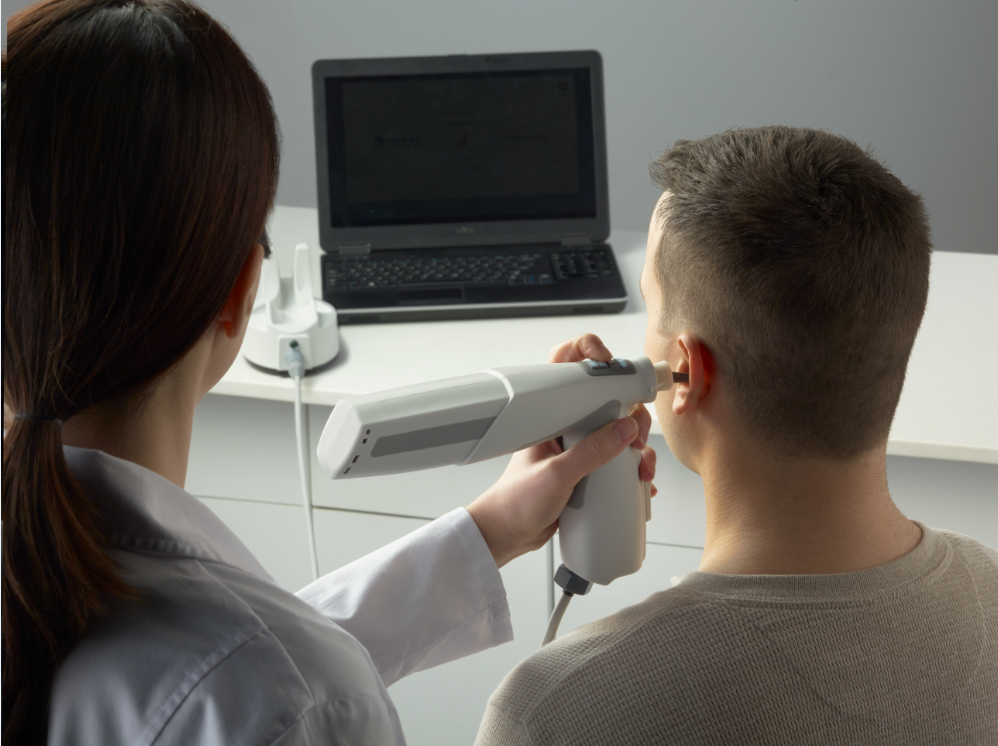
Combatting hearing impairment
The WWH and 3DP4ME have set the objective of producing 12,000 hearing aids to 4,000 individuals in need of hearing aids. According to the partners, whilst the molds for the hearing-aids are 3D printed, the technology inside the device remains the same.
The ultimate goal of this project is to enable children who are hard-of-hearing to enrol in mainstream education and improve the potential for future employability. Furthermore, the Hearing Express Project will seek to facilitate inclusion, employment and full participation in society by elderly persons who are also hard-of-hearing.
This is said to dramatically boost the hope of living a functional life for children and adults, “enabling them to better contribute to the wealth of the nation.” As such, 3DP4ME intends to raise $200,000 for its lab, staff, materials, and training.
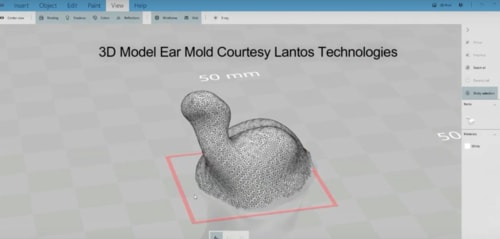
Vote for the 2019 3D Printing Industry Awards.
Subscribe to our 3D printing newsletter and follow us Facebook and Twitter for the latest news in the field of additive manufacturing.
Visit our 3D Printing Jobs board to find out more about opportunities in additive manufacturing.
Featured image shows the Lantos Technologies, 3D Ear Scanner. Photo via Lantos Technologies/3DP4ME.
Tangible Solutions, an Ohio-based manufacturer of medical devices, has validated the quality and consistency of its 3D printed titanium spinal implants. The results of this validation were published in a recent study.
“Additive manufacturing has transitioned from a prototype-only mentality to production-centric technology and as a valid production choice, it begs the question: How reliably does the technology produce a wide mixture of parts?” said Matt Shomper, Director of Engineering at Tangible Solutions.
“Setting up runs of implants in a true production environment is important to ensure consistency. This study has shown that an additive process has the ability to produce process metrics equivalent to manufacturing standards that have existed for decades.”

Assessing 3D printed medical implants
Founded in 2013, Tangible Solutions offers end-to-end management of device manufacture, using additive manufacturing. The company currently provides 3D printing, scanning, and engineering design services. As of 2016, it expanded its metal 3D metal printing capabilities with five Mlab cusing machines and one M2 cusing machine from Concept Laser.
Earlier this year, Tangible Solutions announced the expansion of its facility in Fairborn, Ohio site in order to meet the growing demand of 3D printed medical implants. Now, as part of an internal study, Shomper has analyzed the process capability of its spinal cage implants from a recent alpha launch production run that utilized an estimated 2500 parts.
“Each decision made – whether during the additive design process, setting up for downstream milling, or evaluating critical features and inspection parameters – is carefully considered,” added Shomper.

Keeping up consistency
Data retrieved from the implants indicate that all dimensions of the 3D printed parts maintain tight control over repeatability. As Shomper observes, “The consistency of the process still remains.”
It is also noted that a minority of dimensions were prone to center shifting. According to Shomper, this allows the manufacturer to identify trends and adjust accordingly to keep the models as close to centered, increasing process consistency.
“A process can be ‘in control’ and produce out-of-spec parts. The process may have a high degree of repeatability and consistency but if not mean-centered on the expected tolerance all parts will fail downstream quality checks.”
“Evaluating process capability is understanding the metrics with respect to the customer requirements in terms of limits of the parameters.”
Vote for the best “Medical, dental, or healthcare application” for the 2019 3D Printing Industry Awards.
Subscribe to our 3D printing newsletter and follow us Facebook and Twitter for the latest research in the field of additive manufacturing.
Visit our 3D Printing Jobs board to find out more about opportunities in additive manufacturing.
Featured image shows a 3D printed cage implant. Photo via Tangible Solutions.