Austria-based oral implantologist, researcher and inventor Dr. Mario Kern has developed the Extended Anatomic Platform (EAP), a patented process for creating metal 3D printed dental abutments.
An abutment is used as a connecting element for bridges, denture, and implants. With the integration of GE Additive’s Dental Hybrid Solution, which combines metal 3D printing and CNC milling, Dr. Kern’s EAP aims to “revolutionize dental implant prosthetics.”
“The Dental Hybrid process enables economical production of my abutment with the highest accuracy. With its dental expertise and technical solutions, GE Additive is the perfect partner for my business,” added Dr. Kern, who is also a Professor at the University for Digital Technologies in Medicine and Dentistry (DTMD) in Luxembourg.
“This solution combines the advantages of additive manufacturing with subtractive technology, to get the best out of both manufacturing worlds, which means a time and cost-efficient production process.”
Additive manufacturing and dental implantology
According to Dr. Kern, an increasing amount of patients are being fitted with dental implants; this has resulted in the increased presence of peri-implantitis, an infectious oral disease. More specifically, peri-implantitis inflames the gums and bone structure surrounding a dental implant, causing the tissue around the implant to recede. As a result of this, the metal abutments become exposed.
To remedy this problem, dental technicians use ceramic abutments to cover the exposed metal. This is not a permanent solution, as the ceramic can easily become loose. Therefore, the EAP was created to produce more biocompatible abutments with a natural aesthetic.
Previously, biocompatible, 3D printed dental implants were autonomously placed in the mouth of a volunteer patient by the world’s first autonomous surgical robot in China.
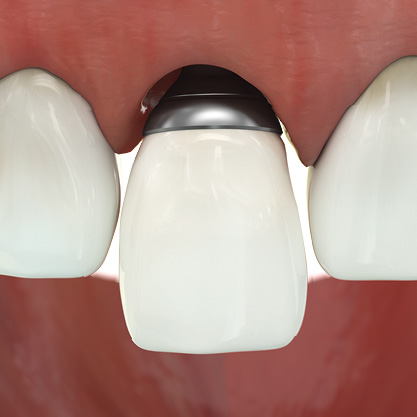
EAP dental abutments
GE Additive’s Dental Hybrid Solution uses Follow-Me open software for data preparation and control of both the Concept Laser Mlab cusing 200R 3D printer and the Datron C5/D5 5-axis milling and post-processing machine.
Dr. Kern used the Mlab cusing 200R to produce narrow wall structures within his abutments. The Datron C5 was then used to further shape these structures. A 3D printable titanium edge is also incorporated in the abutments designed on EAP. This feature is modifiable and eliminates the issue of using adhesive bases for the connecting implant.
The surface of such abutments has been made for optimal cell attachment. “A smooth surface makes cell attachment difficult, and a rough surface facilitates bacterial growth. A roughness of 0.2µm of the EAP surface is ideal as it allows a positive impact on cell growth,” explained Dr. Kern.
With GE Additive’s hybrid dental solution, the EAP process has been said to reduce production costs and time by an estimated 40%. In addition, Dr. Kern reports that abutments manufactured using EAP have a final product density of 99.6%. As such, Dr. Kern has concluded that “additive manufacturing demonstrates better metallurgical properties than with a cast part made from the same material.”
Dr. Kern will launch EAP at the 2019 International Dental Show (IDS) in Cologne, Germany from March 12th-16th.

For more medical and dental 3D printing news, subscribe to the 3D Printing Industry Newsletter, follow us on Twitter, like us on Facebook.
Furthermore, join 3D Printing Jobs for opportunities in additive manufacturing.
Featured image shows the titanium 3D printed EAP abutment. Image via EA-platform.
Researchers from the University of Wollongong (UOW) ARC Centre of Excellence for Electromaterials Science (ACES) in Australia, have developed a customizable 3D bioprinter, 3D Alek, to combat congenital ear deformity.
Designed for the treatment of microtia, the 3D Alek has been installed at the Royal Prince Alfred Hospital (RPA) in Sydney.
“Treatment of this particular ear deformity is demanding because the outer ear is an extremely complex 3D shape, not only in length and breadth but also in height and projection from the skull,” explained Payal Mukherjee, Clinical Associate Professor at the RPA Institute of Academic Surgery (IAS).
“This is where bioprinting is an extremely exciting avenue, as it allows an ear graft to be designed and customized to the patient’s own face using the patient’s own natural tissue – resulting in reduced operating time and improved cosmetic outcome – and avoids the current complication of requiring a donor site for cartilage, usually from the patient’s rib cage.”

3D printed ears
As a genetic condition, microtia affects children and can cause hearing loss. According to Australian charity FutureHear, the disorder “occurs in 1 in 6,000 births world wide” and, as such, there are a number of institutions across the nation are turning to 3D printing to help.
Researchers from UOW and ACES, as well as the Australian National Fabrication Facility (ANFF) created the 3D Alek to make structures using stem cells-containing bioinks, that would aid in cartilage regeneration. Professor Gordon Wallace, Director of ACES, explains, “We have been responsible for the primary sourcing of materials; the formulation of bio-inks and the design and fabrication of a customized printer; the design of required optimal protocols for cell biology; through to the final clinical application.”
“With one 3D Alek now established in a clinical environment at RPA and a replica in our lab at TRICEP, our new 3D bioprinting initiative, we will be able to fast-track the next stages of our research to deliver a practical solution to solve this clinical challenge.”

Pioneering 3D Bioprinting
In addition to TRICEP, Professor Wallace has worked in a number of other notable 3D bioprinting projects. Through ACES and UOW, the University of Sydney’s Save Sight Institute was awarded $1.15 million for the commercial development of its cornea-correcting iFix Pen, last year.
Prior to this, the UOW Intelligent Polymer Research Institute (IPRI), also directed and founded by Professor Wallace, has been using 3D bioprinting to reproduce brain cells, in a project studying conditions such as schizophrenia, epilepsy, and depression.
Having proved 3D Alek in a clinical environment, the next step of UOW and ACES research is to pursue clinical trials, and to develop the ability to work with a patient’s own stem cells.
For more of the latest 3D printing news, follow us on Twitter, like us on Facebook and subscribe to our 3D Printing Industry Newsletter.
Join 3D Printing Jobs for the latest academic opportunities.
Featured image shows UOW researchers with the 3D Alek bioprinter. Photo via UOW.
Kijenzi, a humanitarian start-up from Pennsylvania State University’s College of Engineering, is aiding remote healthcare facilities in Kenya with 3D printed medical supplies.
The initial vision of Kijenzi aimed to use its portable, custom 3D printers to speedily produce health equipment such as braces, clamps and vacuum pumps at low costs. Nevertheless, as the initiative began working in the Kenyan community, the team concluded that access to the CAD design files needed to print the equipment was more limited than the hardware.
Thus, the start-up shifted from supplying 3D printers to developing a cloud-based system, enabling access to 3D printable files for hospitals and medical professionals. John K. Gershenson, co-founder of Kijenzi and director of the Penn State Humanitarian Engineering and Social Entrepreneurship (HESE) program explained:
“Our customers are health care facilities that don’t have access to the supply chains they need and over 40% don’t have the equipment needed to treat their patients. We bring the ability to locally manufacture what they need, when they need it. That is a game changer for access to treatment.”
3D printing with a purpose
Gershenson and Benjamin Savonen, a doctoral candidate in mechanical engineering, co-founded the Kijenzi venture with help from various entrepreneurial support programs at Penn State – particularly, the Penn State HESE program. This challenges students and faculty to create technology-based solutions that will positively affect those in lower income communities using sustainable business models.
Kijenzi was further developed through its participation in the Ben Franklin Technology Center’s TechCelerator program. Gershenson added, “The TechCelerator gave us the time and feedback we needed to craft the story of Kijenzi in such a way that everyone could understand what we are doing.”
Savonen identified the need for new lab equipment while teaching in Africa. This led the venture to rural areas of Kenya. 3D printing was recognized as an appropriate solution to manufacture hard-to-get items such as anatomical models, replacement parts, and occupational therapy devices – which contribute to the well-being of others.

A sustainable supply chain
In many cases, supply chains can be limited in its reach as a result of broken and obsolete machinery, as well as logistical difficulties. Kijenzi has addressed these limitations with its cloud-based system which holds approximately 400 parts in its inventory.
The 3D printed equipment has been tested and used within collaborative educational institutes as well as medical facilities. Kijenzi is now preparing to launch an initial program in Kenya this May.
Similarly, last year, Field Ready, a non-profit organization based in Illinois, addressed the logistical challenges, medical, and social needs of the Sharia Camp in Iraq with novel soap with 3D printed toys inside.
Nominate Penn State’s Kijenzi among others while you still can for the upcoming 2019 3D Printing Industry Awards.
For further awards updates and ideas for who to nominate this year, subscribe to the 3D Printing Industry newsletter, follow us on Facebook and like us on Twitter.
Seeking 3D Printing Jobs? Join and advertise on our dedicated site now to reach professionals in this industry.
Featured image shows 3D printed tweezers. Clip via Kijenzi/Penn State.
ROKIT Healthcare, a Korean 3D bioprinter manufacturer previously known as ROKIT, has introduced a novel 3D bioprinting method to treat scarred lesions.
Using the company’s 3D bioprinter, the ROKIT INVIVO, to 3D print a patient’s autologous tissues and cells into a dermal patch graft. When placed on a wound, the 3D printed dermal patch graft enables the natural formation of new blood vessels (known as neovascularization) for effective regeneration of the skin.
“It is a novel way of overcoming some of the most pronounced limitations associated with traditional stem cell therapy,” said Seok-Hwan You, Chief Executive Officer of ROKIT Healthcare.
“Utilizing 3D bioprinting techniques allows for effective delivery of autologous cells unto the wound site, minimizing cell loss and greatly enhancing cell viability and proliferation.”

3D bioprinting to heal wounds
According to You, “The current practice of directly injecting expanded or cultured stem cells using syringes severely limits cell viability and accurate, uniformly distributed delivery of effective stem cells unto the disease area.”
This hinders treatments for skin scarring, one of the most common medical conditions following dermal injuries, the company states. Such scars can cause significant physical, aesthetic, psychological and social issues, resulting in elective surgical procedures.
To address these limitations, ROKIT Healthcare has been working since 2012 in regenerative medicine to develop additive manufacturing solutions for personalized and improved patient care. ROKIT’s method leverages 3D bioprinting technology as it allows for uniform and equal-density distribution of patient’s autologous tissue and cells.
This leads to rapid migration of keratinocytes – cells designed to form tight junctions with the nerves of the skin – which enables neovascularization at the site of a wound. The company plans to evaluate the effectiveness of this treatment in clinical studies to expand its application to other dermatological conditions including burns and ulcers.

3D bioprinting and regenerative medicine
ROKIT Healthcare’s research into in-situ bioprinting for skin regeneration prompted the release of the Edison Invivo 3D Bioprinter in 2016. This was made possible from a $3 million grant from the Korean Government’s Ministry of Strategy & Finance.
Similarly, skin regeneration and 3D bioprinting have also been used by researchers from Shanghai Jiao Tong University, the National Tissue Engineering Research Center of China, the Chinese Academy of Medical Science, Wei Fang Medical College and Dalian University, in China to constructed ears for children suffering from microtia.
Don’t forget to nominate ROKIT Healthcare and others for the upcoming 2019 3D Printing Industry Awards.
For further awards updates and ideas for who to nominate this year, subscribe to the 3D Printing Industry newsletter, follow us on Facebook and like us on Twitter.
Seeking 3D Printing Jobs? Join and advertise on our dedicated site now to reach professionals in this industry.
Featured image shows patient cells inside the ROKIT INVIVO’s syringe. Clip via ROKIT Healthcare.
Additive manufacturing and 3D scanning have been increasingly integrated within the healthcare sector. 3D Systems, in particular, has contributed significantly to the advancement of surgical procedures with this technology.
Last year, the company, based in Rock Hill, South Carolina, began offering its Virtual Surgical Planning (VSP) technology as well as its range of 3D anatomical models through medical technology firm Stryker. As a result, 3D Systems has provided VSP or anatomical services in more than 100,000 cases, including facial reconstruction surgery and full facial transplants.
But in addition to this AR/VR is also used by 3D Systems to help medical professionals improve surgical outcomes.
Insights from one VP at company show how 3D printing is not the only technology addressing the healthcare sector at 3D Systems.
Ran Bronstein has worked in the field of medical simulation for over twenty years and co-founded Simbionix, a company providing 3D virtual reality surgical simulation, which was acquired by 3D Systems in 2014. With his experience, Bronstein aims to leverage anatomical modeling and simulation technologies, to train medical professionals for better patient-specific based healthcare.
In a recent interview for The Jerusalem Post Bronstein, 3D Systems Vice President, and Chief Research and Operations Officer, commented, “Physicians can practice on animals or on cadavers, and sometimes need to practice on actual patients. We want them to be very well trained, and computerized simulation is something that aims to replace these old methods.”
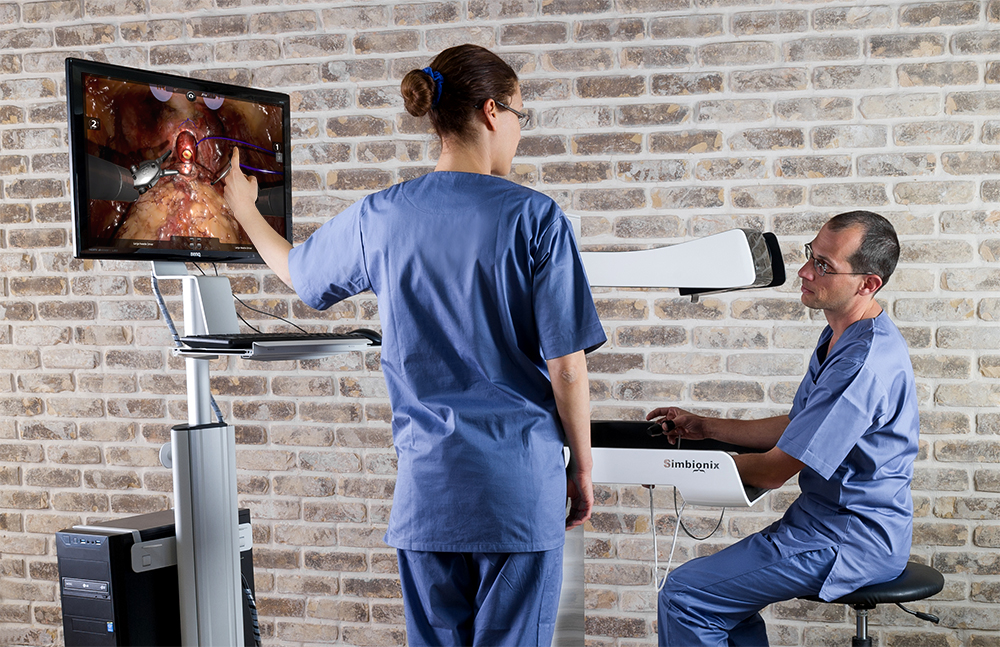
A simulation for surgical success
“Pilots have been using simulators for many years. But there are more physicians than pilots,” stated Bronstein. “Sometimes physicians make a greater number of mistakes than pilots. To prevent these mistakes and improve patient safety and procedure outcome, we decided to develop a medical simulator based on the same concept.”
Among its simulation solutions is the RobotiX Mentor training module. This allows trainees to practice surgeries in a virtual environment using step-by-step guidance, performance metrics, anatomy recognition, and a video-based curriculum.
“The operating room is becoming more and more complicated, with more modules: virtual reality, X-Ray, ultrasound, MRI, CT and robotics. It is becoming an increasingly technological and complicated environment. Without a simulator, there will be no ability to carry out surgery.”
3D printing helps future physicians
In addition to virtual reality, 3D Systems has also added to its medical portfolio through its recent partnership with non-profit organization OpHeart and its Heart-in-Hand Pledge. More specifically, the company is using its D2P (DICOM to Print) software and ProJet CJP 660Pro ColorJet 3D printers and VisiJet materials to produce customized 3D printed heart models for doctors tending to young patients with Coronary heart disease (CHD).
Prior to this, 3D Systems supported the landmark surgical case of the conjoined McDonald Twins and manufactured over 600,000 3D printed medical devices. The company also recently launched an On Demand Anatomical Modeling Service. This service makes custom 3D printed models to be used in surgical planning and patient education.
Nominate 3D Systems and others for the upcoming 3D Printing Industry Awards 2019.
Want the latest 3D printing news from the healthcare sector? Subscribe to the 3D Printing Industry newsletter, follow us on Twitter and like us on Facebook.
Looking for a fresh start this year? Visit 3D Printing Jobs to commence your career in additive manufacturing.
Featured image shows a CJP pediatric heart model Photo via 3D Systems.
Orchestrate Orthodontic Technologies (O3D), a Californian dental digital solutions provider, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Orchestrate 3D Treatment Planning Software System, which includes 3D printing and clear aligner fabrication.
“There is no question that 3D printing is rapidly changing the field of orthodontics,” said Todd Ehrler, DDS, MS, founder, Orchestrate Orthodontic Technologies.
“The Orchestrate 3D treatment planning software and 3D printing solutions provide the practitioner with turn-key solutions to in-source aligner therapy and other orthodontic appliances.”
“Our host of products, services, and comprehensive training allow the practice to quickly integrate orthodontic-specific 3D printing technology and software.”
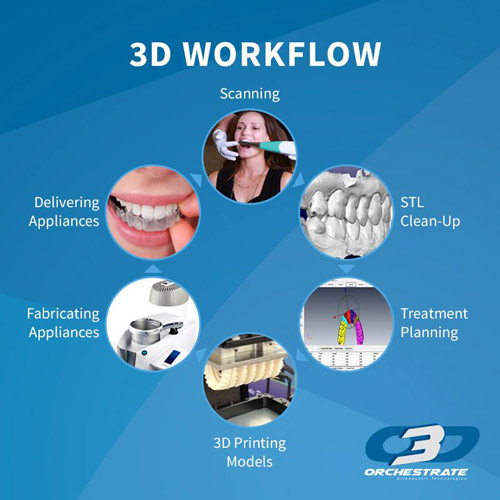
The Orchestrate 3D Treatment Planning Software System
Founded in 2007, O3D is formed from experienced orthodontics adapting new technologies specifically for dentistry. On its journey to develop an advanced “in office” 3D digital orthodontic system, the company partnered with EnvisionTEC, a global 3D printer, and materials manufacture.
In the past five years, O3D tested and researched the 3D scanners, software, and 3D printers to integrate into the Orchestrate 3D Treatment Planning Software System. As such, O3D partnered with Park Dental Research in New York to produce its own dental SLA 3D printer, the Juell O3D-2.
According to O3D, the newly approved software system is also compatible with a range of scanners and additive manufacturing systems. “If your scanner produces an STL file our O3D software will accept it.”
The 510(K)clearance now enables dental professionals in the U.S. to use the Orchestrate 3D Treatment Planning Software System to design and produce their own orthodontic clear aligner appliances.

The software suite is capable of case analysis, setup and staging, and exporting to print for production for clear aligners. Some of the functions include analysis of overjet/overbite, collision detection, occlusion, and spaces.
Following the case analysis, virtual setups are created and subdivided into the clear aligner stages needed. When planning is complete, sub-setups can be prepared for additive manufacturing. Furthermore, the software is open, allowing users to decide how much of the workflow they want to do in-house or from production partners.
Additionally, the treatment planning and simulations can be shared onscreen with patients, the dental practice and lab, via am online web viewer.
3D printing and the dental sector
Additive manufacturing in the dental sector has been present throughout the past few years. In 2018, Carbon, the Silicon Valley-based company behind Digital Light Synthesis (DLS) 3D printing technology, released its 3D printable DENTCA resins with approval from the FDA.
In a related manner, Stratasys began with working with leading U.S. and Spanish orthodontics research labs, Dynaflex and Ortoplus, to advance the large-scale production of clear aligner molds with the Stratasys J700 Dental 3D Printer.
Similarly, EnvisonTEC announced its FDA approved E-Denture material for 3D printing dentures.
Remember, nominations for the 3D Printing Industry Awards 2019 are open. Let us know the medical enterprises, technology and individuals leading the additive manufacturing sector.
For more updates on Vader Systems acquisition by Xerox, subscribe to the 3D Printing Industry newsletter, follow us on Twitter and like us on Facebook.
Why not visit 3D Printing Jobs to commence your career in additive manufacturing
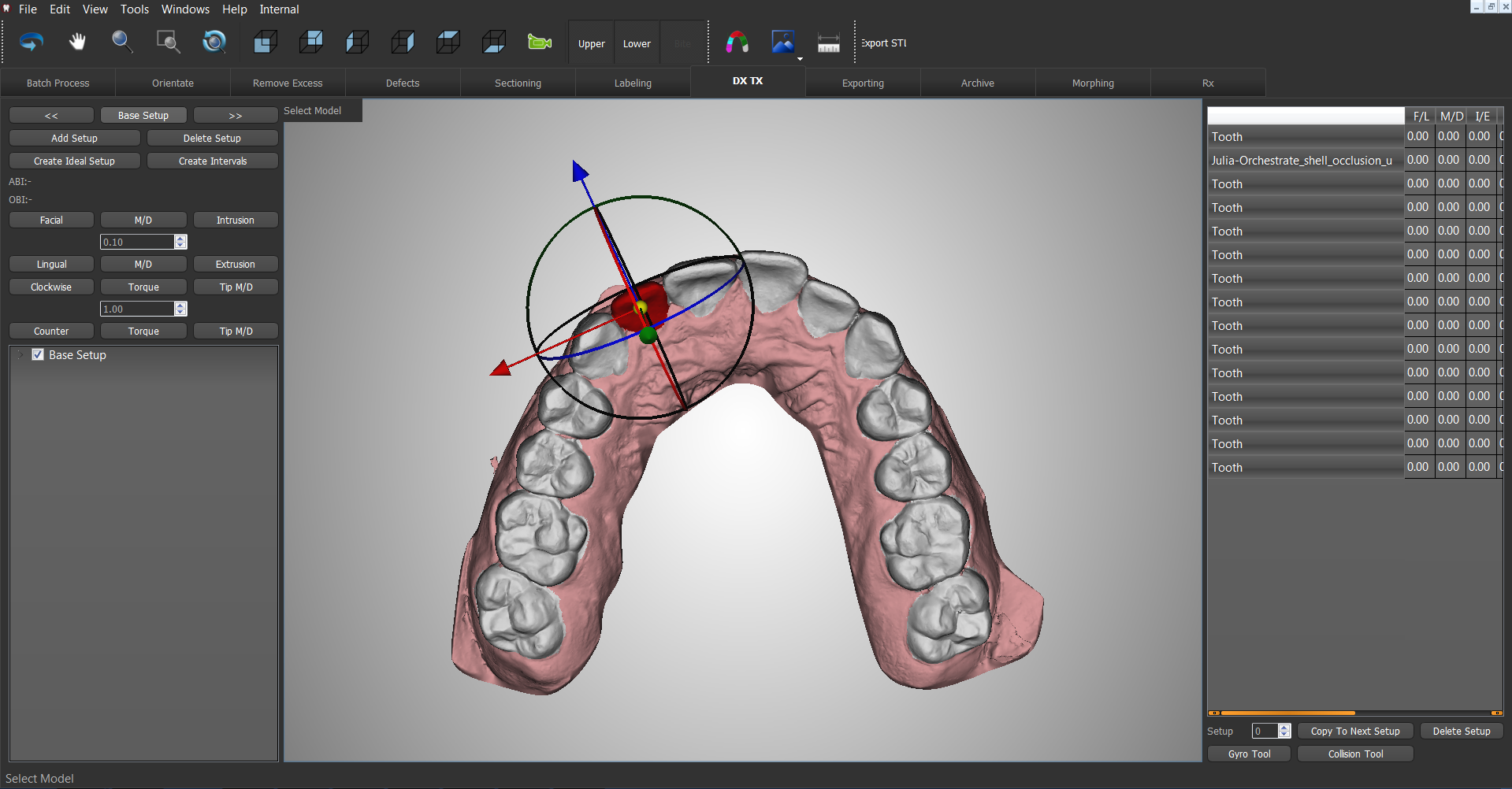
Featured image shows a 3D dental scan. Image via O3D.
Carbon, a California-based 3D printer and materials manufacturer, has partnered with Dentsply Sirona, a Pennsylvania-based manufacturer of dental equipment, to produce novel denture materials.
Such materials will be processed using Carbon’s Digital Light Synthesis technology to create high-quality 3D printed dentures.
“We’re very excited to team with Carbon to deliver a total seamless workflow for increasing laboratory efficiency and elevate state-of-art materials for removable appliance fabrication in the 3D dental printing space,” said Julie Mroziak, Vice President of the Global Lab Portfolio, Dentsply Sirona.
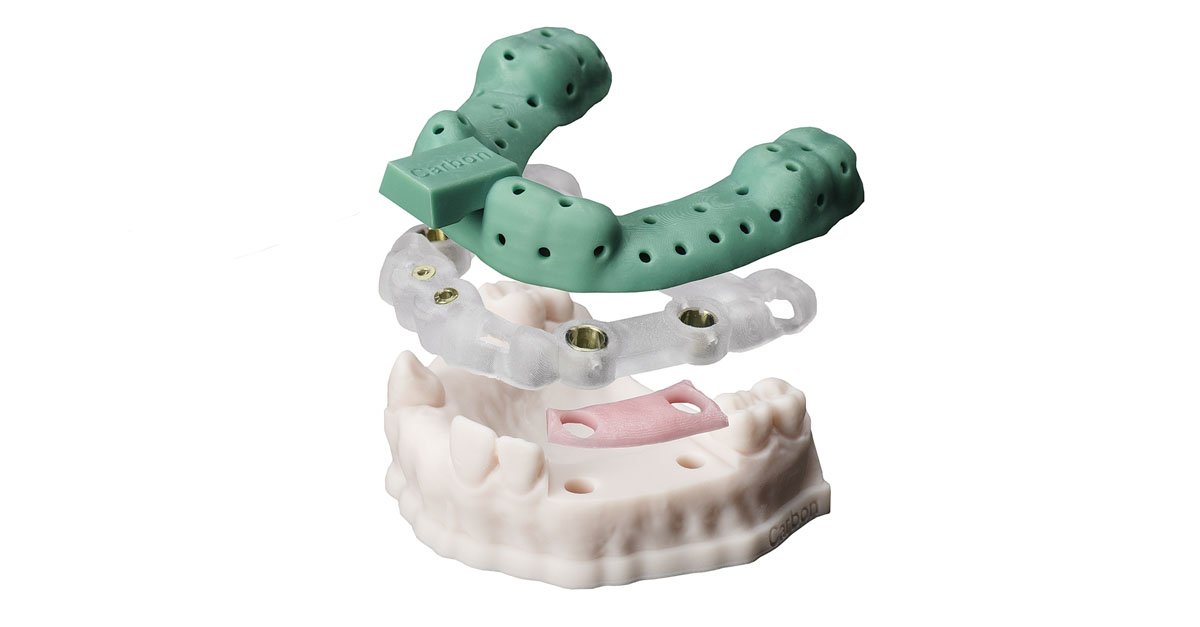
Accelerating 3D printing in the dental market
With over 100 years of experience in the dental market, Dentsply Sirona has developed a wide range of dental equipment including mouth guards, retainers, orthodontic implants, and denture liners.
The company also has expertise in scanning and CAD/CAM software and is a partner of the new Massachusetts Institute of Technology (MIT) Center for Additive and Digital Advanced Production Technologies (ADAPT).
Through this collaboration, Dentsply Sirona will leverage DLS technology to develop materials with improved strength, aesthetics, and laboratory efficiencies for 3D printed dentures.
Digital Light Synthesis and the medical sector
DLS technology uses light and oxygen to rapidly produce high-quality, end-use products from a pool of resin, an alternative method to stereolithography (SLA). This process works in tandem with Carbon’s high-performance materials and is enabled by continuous liquid interface production (CLIP), a photomechanical process with a liquid interface of uncured resin and a passing light source.
Several institutions are exploring DLS and its capability to produce medical and dental devices. Earlier this month, Grand Valley State University (GVSU) and the Applied Medical Device Institute (aMDI) in Michigan received $500,000 to create production-grade medical parts with the Carbon M2 3D printer. Furthermore, Carbon recently introduced its first medical-grade material: Medical Polyurethane 100 (MPU 100).

Digital denture solutions
Brian Ganey, General Manager of Carbon’s Dental Business commented on the recent collaboration with Dentsply Sirona, “This partnership with Dentsply Sirona is a testament to Carbon’s global vision and mission to enhance the digital ecosystem. Together, we will be able to offer the marketplace a complete digital denture solution that is superior in terms of strength and aesthetics.”
“With Carbon as the leading digital manufacturer in digital dentistry and Dentsply Sirona’s global leadership in removable appliances, we can deliver an unmatched offering that benefits both the dental laboratory and the patient.”
The Dentsply Sirona denture materials for Carbon digital manufacturing solutions will be available in the U.S. later this year.
It’s not too late to nominate for the upcoming 3D Printing Industry Awards 2019.
Want the latest medical and dental 3D printing news? Subscribe to the 3D Printing Industry newsletter, follow us on Twitter and like us on Facebook.
Looking for a fresh start this year? Visit 3D Printing Jobs to commence your career in additive manufacturing.
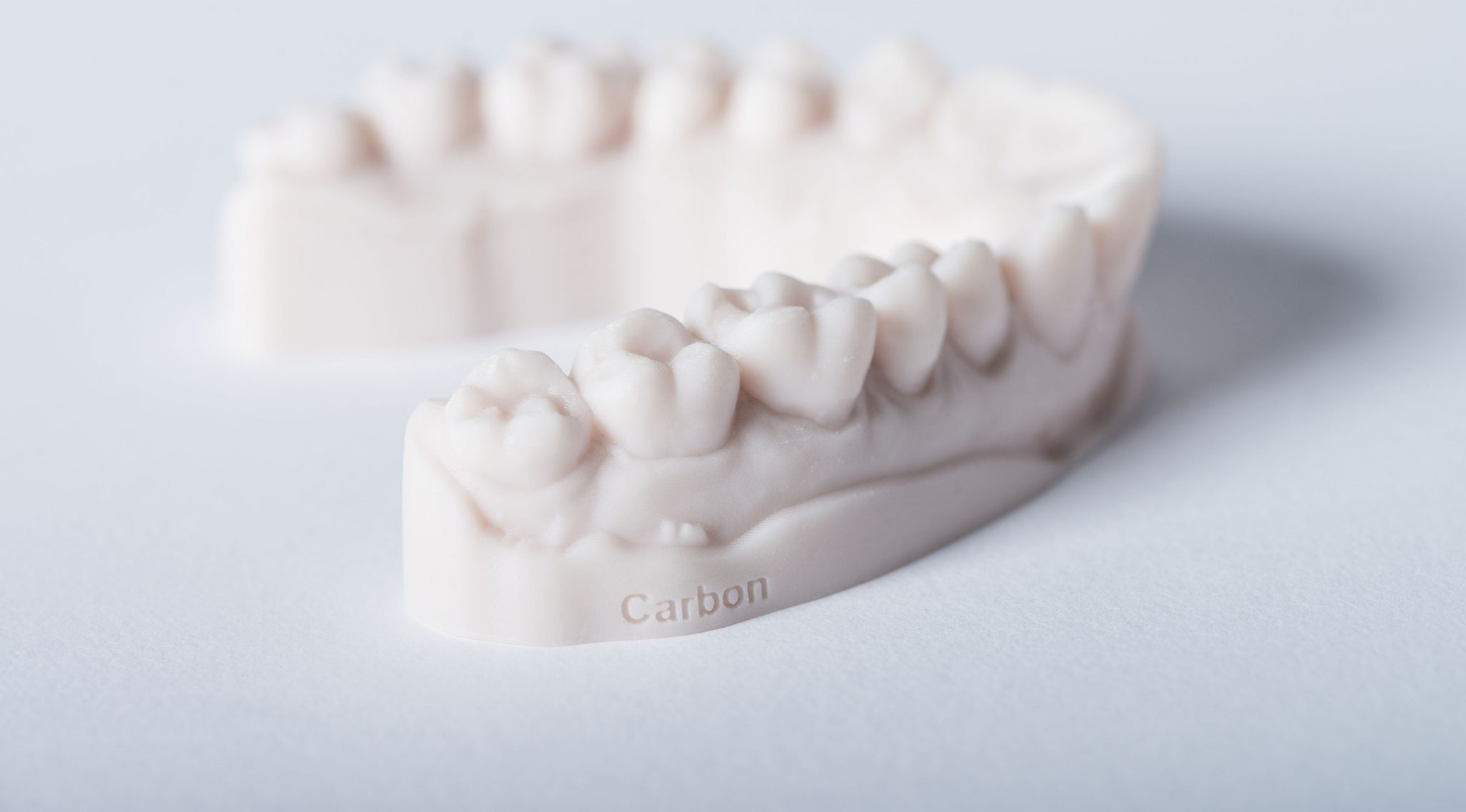
Featured image shows a 3D printed dental impression. Photo via Carbon.
Researchers from the University of California San Diego (UCSD) School of Medicine and Institute of Engineering in Medicine (IEM) have successfully 3D printed a two-millimeter spinal cord implant to repair spinal cord injuries in rats.
In yesterday’s issue of Nature Medicine, the researchers explained in their study that the 3D printed implant contains 200-micrometer-wide channels (twice the width of a human hair) that guide neural stem cell growth, ultimately restoring connections and lost function.
“This shows the flexibility of our 3D printing technology,” said co-first author Wei Zhu, PhD, a nanoengineering postdoctoral fellow in the Chen group.
“We can quickly print out an implant that’s just right to match the injured site of the host spinal cord regardless of the size and shape.”
3D bioprinting and vascularization
Vascularization is the process in which blood vessels are formed. According to Zhu, it is “one of the main obstacles in engineering tissue implants that can last in the body for a long time.”
“3D printed tissues need vasculature to get enough nutrition and discharge waste. Our group has done work on 3D printed blood vessel networks before, but we didn’t include it in this work. Biology just naturally takes care of it for us due to the excellent biocompatibility of our 3D scaffolds.”
In December, UCSD bioengineers developed a 3D bioprinting method that integrates natural materials which produce lifelike organ tissue models. Prior to this, the researchers gained experience in bioengineering by 3D printing a framework of functional blood vessels using a Digital Light Processing (DLP) method with hydrogel and encapsulated cells.

A 3D printed spinal cord implant
The 3D printing technology used by the researchers produced the two-millimeter-sized implants in 1.6 seconds. In comparison, the study states that traditional nozzle printers take several hours to produce much simpler structures.
Loaded with neural stem cells, the team grafted the implants into sites of severe spinal cord injury in rats. In a few months, new spinal cord tissue had regrown across the injury and connected the severed ends and axons (long, threadlike extensions on nerve cells) of the host spinal cord.
This experiment resulted in the treated rats regaining significant functional motor improvement in their hind legs. Following this, the researchers proved this process was scalable to human spinal cord sizes by 3D printing four-centimeter-sized implants modeled from MRI scans within 10 minutes.
“This marks another key step toward conducting clinical trials to repair spinal cord injuries in people,” said Kobi Koffler, PhD, Assistant Project Scientist at the Center for Neural Repair, UCSD.
Mark Tuszynski, MD, PhD, professor of neuroscience and director of the Translational Neuroscience Institute at UC San Diego School of Medicine, concluded:
“We’ve progressively moved closer to the goal of abundant, long-distance regeneration of injured axons in spinal cord injury, which is fundamental to any true restoration of physical function.”
The research paper “Biomimetic 3D printed scaffolds for spinal cord injury repair,” is co-authored by Xin Qu, Oleksandr Platoshyn, Jennifer Dulin, John Brock, Lori Graham, Paul Lu, Martin Marsala, and Jeff Sakamoto.

Nominate for the upcoming 3D Printing Industry Awards 2019.
For updates on the latest 3D printing medical research, subscribe to the 3D Printing Industry newsletter, follow us on Twitter and like us on Facebook.
Looking for a fresh start this year? Visit 3D Printing Jobs to commence your career in additive manufacturing.
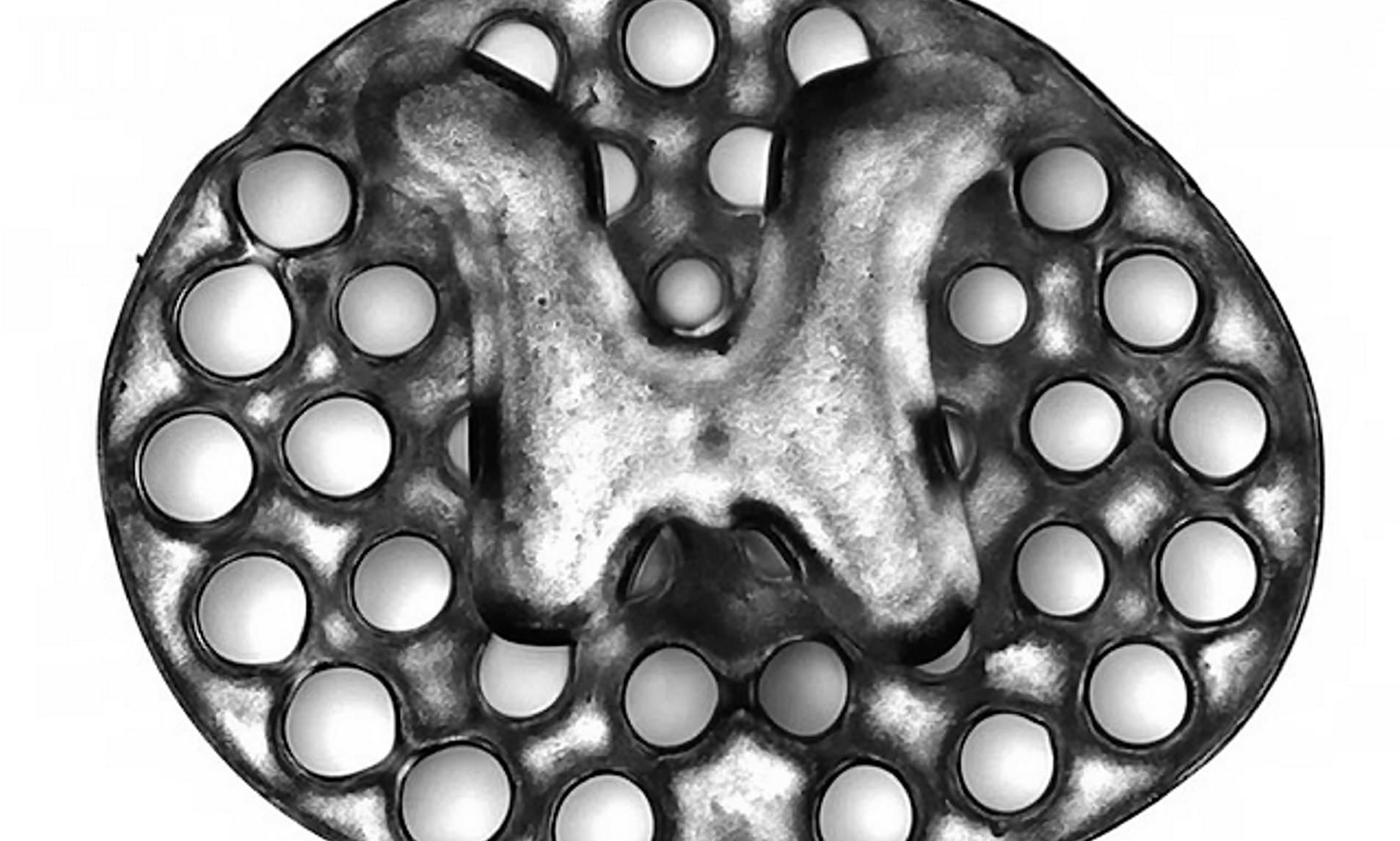
Featured image shows the 3D printed implant used as scaffolding to repair spinal cord injuries in rats. Image via Jacob Koffler/Wei Zhu, UCSD.
Korea University Guro Hospital (KUGH) and the Korea National University of Transportation’s (KNUT) 3D Printing Chungbuk Center have launched a program which seeks to use additive manufacturing to aid patients with fractures.
Initiated by South Korea’s Ministry of Science and ICT(MSIT), the program aims to provide surgeons with patient-specific 3D printed anatomical models. Such models will address complications experienced after fracture surgery with minimally invasive surgeries. Professor Oh Jong-gun of the department of orthopedics at KUGH explained:
“The deformation of the bone often leads to complications in fracture surgery patients due to repeated surgery and the fracture site [i.e.,] nonunion and malunion. This makes operation very difficult as the conventional anatomical plate does not fit the complication.”
“However, the team has found [that] 3D printing technology can provide a breakthrough solution for the treatment for such fracture complications.”
Treating fractures with 3D printing
Injured bones may heal by with a cast, however, severe fractures require more invasive treatments, such as bone fracture repair surgery, which uses metal screws, pins, rods, or plates to hold the bone in place.
According to Professor Oh Jong-gun, “patients who had complications after fracture surgery have had a hard time using existing metal plates, as it did not perfectly match their injury.”
“Such issues led the surgeons to bend the metal plate by hand during operations, which increased operation time and resulted in re-operation in some cases.”
The KNUT will support the KUGH in 3D printing anatomical models of a patient’s bones based on the individual’s CT and MRI image data. This process will enable surgeons to form contours in the metal plates in advance to fit the patient’s bone shape.
2D and 3D images of bones are usually all the resources surgeons have to perform a procedure. With 3D printing, the medical professional can simulate the process beforehand with physical models, and use them as templates throughout.
Ultimately, the institutions hope to increase minimally invasive operations with significantly reduced operation time.

South Korea accelerate additive manufacturing
The Government institution MSIT is centered on the overall adoption of 4th Industrial Revolution technologies within Asia. Previously, the MSIT invested $5.7 million the Electronics Telecommunications and Research Institute (ETRI) who is developing a 3D scanning smartphone application.
Following this, the MSIT announced it would be spending 41.2 billion won (Approx. $37 million USD) of 2017’s annual budget to encourage the development of 3D printing across the nation.

Nominate for the upcoming 3D Printing Industry Awards 2019.
For the latest medical 3D printing news, subscribe to the 3D Printing Industry newsletter, follow us on Twitter and like us on Facebook.
Seeking a fresh start in the new year? Visit 3D Printing Jobs to commence your career in additive manufacturing.
Featured image shows the Korea University Guro Hospital (KUGH). Photo via KUGH.
ActivArmor, a Colorado-based manufacturer of 3D printed orthoses, an external device which supports the limbs or spine, has expanded its operations as a result of its partnered with Jacksonville Orthopaedic Institute (JOI).
Made from high-temperature thermosetting plastics – similar to LEGO – ActivArmor devices are waterproof and customized to suit the patient. According to the company, the lattice design of the orthosis can be molded to accommodate post-surgical hardware, incisions, scars, and burns.
“JOI’s expertise in sports medicine makes them an ideal provider of this next-gen product,” explained Diana Hall, Founder of ActivArmor.
“Being right on the field with their patients, they are able to see the benefits of our hygienic, waterproof orthoses first-hand, and provide their patients with the latest in custom care solutions to improve healing outcomes and quality of life, both on and off the field.”

A breathable, 3D printed cast
Based in Florida, JOI is the official Sport Medicine Provider for teams including the Jacksonville Armada FC, the Blue Wave Swim Team, and the Jacksonville Jaguars football team. As a result of the partnership with ActivArmor, JOI’s 34 board-certified physicians can now offer ActivArmor products to their patients.
With the help of 3D mapping, ActivArmor devices are created to fit a patient’s injured limb. This method is more accurate and adaptive than conventional methods of support such as progressive layers of tape or plaster.
Kevin Kaplan, an orthopedic surgeon at JOI, and head team physician for the Jacksonville Jaguars told The Florida Times-Union:
“The benefits for athletes to be able to sweat, shower, ice and train while healing from an injury are obvious, but ActivArmor is also great for anyone who wants to remain active — from children to post-surgical patients.”
Following 5 years of field-testing on the high-tech waterproof, hygienic, breathable cast/splinting alternative, ActivArmor plans to expand further into the north Atlantic coast.

3D printed orthoses
The ability to customize 3D printed models has been frequently leveraged within the healthcare sector. Students at Gonzaga University in Washington have recently used additive manufacturing technology to produce an Ankle Foot Orthosis (AFO) for children. The brace holds the foot and lower leg in position and helps children develop motor skills and build strength in and around their joint.
Following this, students at Barcelona’s Polytechnic University of Catalonia (UPC) developed a 3D printed orthotic fin for a 16-year-old swimmer who suffered from a stroke and partial paralysis. The fin has aided the young swimmer’s positioning in the water, allowing him to build upper body muscle.
Submit your nominations now for the 3D Printing Industry Awards 2019.
Also, for the latest medical 3D Printing Industry updates subscribe to our newsletter, follow us on Twitter and like us on Facebook.
Seeking a fresh start in the new year? Visit 3D Printing Jobs to commence your career in additive manufacturing.
Featured image shows ActivArmor 3D printed casts. Photo via ActivArmor.