Leading 3D printer manufacturer 3D Systems has taken another step forward in its medical portfolio through a partnership with non-profit organization OpHeart.
Based in Houston, Texas, OpHeart integrates additive manufacturing technologies to provide children born with life-threatening heart defects with the best medical treatment available. As part of this initiative, the company also operates a “Heart-in-Hand Pledge”, granting any requesting doctor or parent with a 3D printed model of their Coronary heart disease (CHD) patient’s heart before surgery, regardless of the family’s or hospital’s ability to pay. In this collaboration, 3D Systems is participating in OpHeart’s “Heart-in-Hand Pledge.”
Anne Garcia, Co-Founder and Executive Director, OpHeart stated:
“It is our hope that as more surgical teams work with OpHeart to employ 3D Systems’ anatomical models in their CHD surgeries, we will be able to definitively demonstrate what is common sense – providing doctors the ability to better prepare for complex surgeries makes a meaningful, measurable difference in the lives of these children.”
3D printed anatomical models
The American Heart Association recognizes CHD as the most common birth defect within the U.S. As a result of CHD, over 100,000 babies are in need of high-risk surgeries in order to survive.
After her own daughter was born with a life-threatening heart defect, Garcia co-founded OpHeart in 2015.
Over the past four years, 3D Systems has leveraged its 20 years of anatomical modeling expertise and medical workflow to help OpHeart produce accurate, detailed 3D printed models of CHD patients for a surgeon’s use. Garcia added:
“We fiercely believe that the ability to 3D print a replica of a CHD patient’s heart is an invaluable tool that can, to put it bluntly, save lives.
“By giving surgeons the ability to practice and plan for complex surgeries that involve reconnecting vessels as thin as human hairs in hearts no larger than a strawberry, it only stands to reason that the quality of surgery improves. We want every child with a life-threatening heart defect to benefit from this technology.”
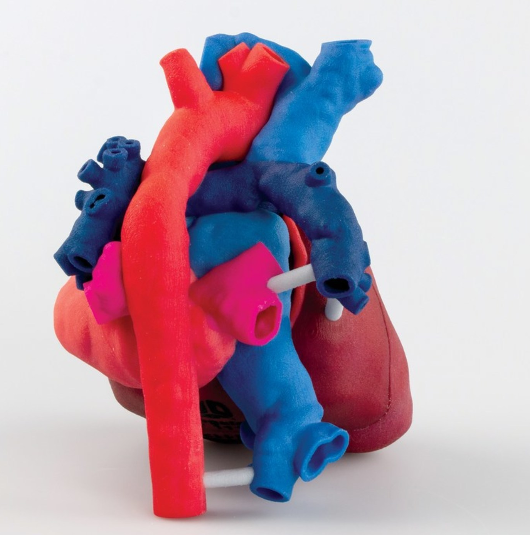
The Heart-in-Hand Pledge
3D Systems is largely contributing to the Heart-in-Hand Pledge by leading the creation of customised heart models. With the use of its D2P (DICOM to Print) software, the company’s team of expert biomedical engineers is converting MRI and CT scans data of a heart into a 3D model. Following this, the model is printed using ProJet CJP 660Pro ColorJet 3D printer and VisiJet materials. The models are then shipped worldwide.
As there is no insurance reimbursement for these tools, 3D System’s charitability intends to accelerate the Heart-in-Hand initiative in saving children’s lives. Garcia concluded, “Hopefully, they will become the standard of care, as insurance companies recognize their value and reimburse for their use.”
For more on additive manufacturing in the medical sector, subscribe to the 3D Printing Industry newsletter. Also, find us on Facebook and like us on Twitter.
Looking for a career change? Search and post new opportunities across additive manufacturing by visiting 3D Printing Jobs.
Featured image shows a 3D printed heart model. Photo via 3D Systems/OpHeart.
Health Canada, the national public health department of the Government of Canada, has released a draft guidance document for manufacturers intending to license implantable 3D printed medical devices.
The document states, “Due to the fast-changing technological environment, Health Canada will continue to adapt its policy approach to 3D-printing as issues on the topic evolve,”
“This guidance document therefore represents the first phase of 3D printing policy in Canada.”
Licensing Requirements for 3D printing medical devices
The draft guidance document remains consistent with the standard definitions from the International Medical Device Regulators Forum (IMDRF). The document states, “hospitals that manufacture 3D printed implantable medical devices for distribution beyond their institution qualify as a manufacturer, and should comply with the same obligations as other medical device manufacturers.”
As a result, such manufacturers are instructed to consider the design philosophy, biocompatibility, performance, market history, and packaging of a 3D printed medical device.
“If the application is to modify a licensed device, a description of the modification is required. Examples of device modifications related to 3D printing may include, but are not limited to [a] 3D printed component that is being added to an approved device (i.e., porous surface coating) [and] a 3D printed component that is to be used with an approved device (i.e., dental superstructures/abutments/implants).”

Safety requirements of 3D printed medical devices
Within the draft guidance document, safety considerations have also been proposed to ensure the effectiveness of 3D printed medical devices. This includes post-processing and sterilization, animal studies and clinical studies, software verification and validation, as well as shelf life studies for the device.
Furthermore, according to the device description policies, manufacturers must state whether 3D printing was used to produce only a part of the component or to produce the device in entirety. The draft guidance is currently seeking stakeholder feedback on the technical considerations outlined until 8 January, 2019.
3D printing for the healthcare sector
The use of additive manufacturing within the medical sector has seen great progress over the past few years. As a result, the U.S. Food and Drug Administration (FDA) Commissioner Dr. Scott Gottlieb released a statement reaffirming the agency’s commitment to a “new era of 3D printing of medical products.” This statement also revealed the FDA’s guidance for manufacturers submitting 3D printed devices for approval. On the announcment Dr. Gottlieb stated:
“3D printing is certain to alter the daily practice of medicine where patients will be treated with medical products manufactured specifically for them.”
Stay updated with the latest additive manufacturing news subscribe to the 3D Printing Industry newsletter. Also, find us on Facebook and like us on Twitter.
For new opportunities across additive manufacturing visit 3D Printing Jobs.
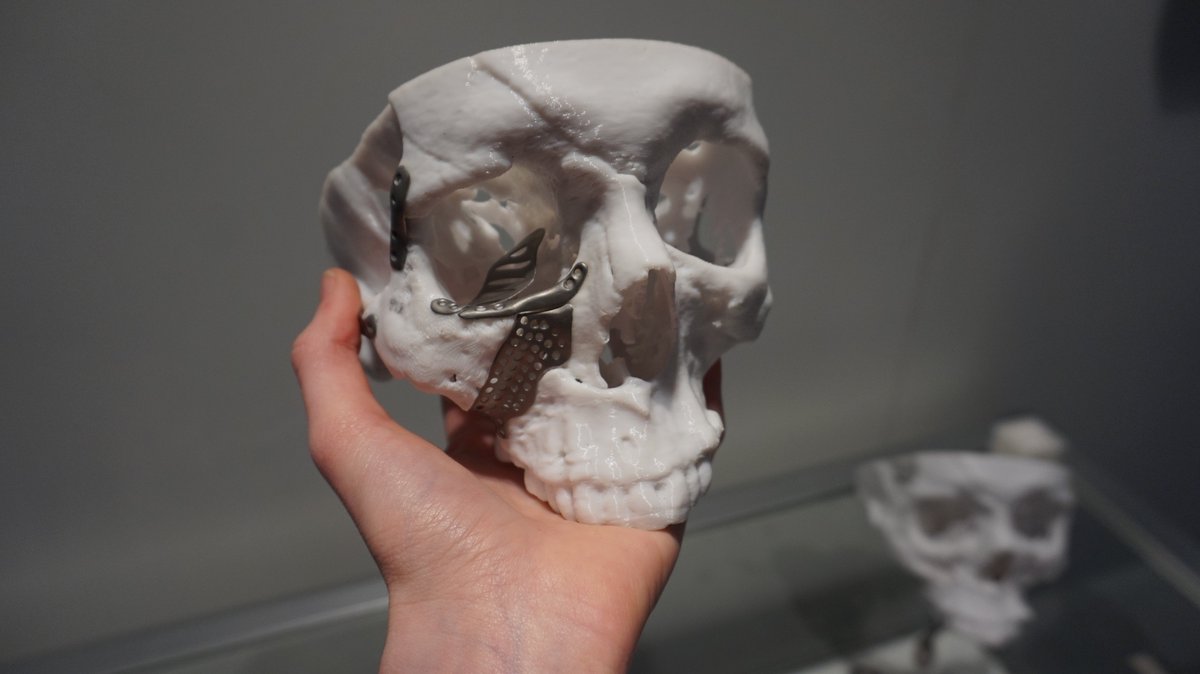
Featured image shows a 3D printed skull and implant by Renishaw. Photo by Beau Jackson.
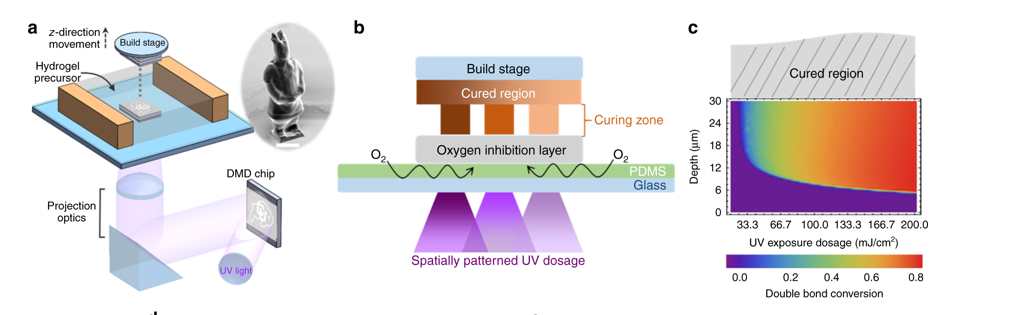
Researchers from the University of Colorado Boulder (CU Boulder) have developed a 3D printing method using controlled oxygen inhibition to imitate the highly structured, pliable geometry of blood vessels.
“The idea was to add independent mechanical properties to 3D structures that can mimic the body’s natural tissue,” said Xiaobo Yin, an Associate Professor in CU Boulder’s Department of Mechanical Engineering and the senior author of the study.
“This technology allows us to create microstructures that can be customized for disease models.”
Hardening 3D printed blood vessels
According to the research, engineering a solution for hardened blood vessels associated with cardiovascular disease and viable artery and tissue replacement has been “historically proven challenging”. Using a 3D printer built with parts purchased from eBay at a total cost of $500, the researchers developed a stereolithography 3D printing method to manufacture microscopic structures.
This method integrates an oxygen inhibition layer between a cured polymer structure to physically limit the curing thickness during the additive manufacturing process.“Oxygen is usually a bad thing in that it causes incomplete curing,” said Yonghui Ding, a Postdoctoral researcher in Mechanical Engineering and the lead author of the study. “We utilize a layer that allows a fixed rate of oxygen permeation.”
“By keeping tight control over oxygen migration and its subsequent light exposure, the researchers have the freedom to control which areas of an object are solidified to be harder or softer—all while keeping the overall geometry the same.”
Mimicking the diseased blood vessels
Testing their concept, the researchers printed three versions of structure featuring a top beam supported by two rods. The structures were identical in shape, size, and materials, but had been printed with three variations in rod rigidity: soft/soft, hard/soft and hard/hard. Following this, the team found that the harder rods supported the top beam while the softer rods allowed it to fully or partially collapse.
The researchers also printed a small Chinese warrior figure, with hard outer layers and soft interior layers, which gave the warrior varied textures. CU Boulder believes that this method is “opening a new avenue towards 3D in vitro tissue fabrication.” This research is also said to accelerate more personalized treatments for those suffering from hypertension and vascular diseases.
“We were able to create this structure to mimic the diseased blood vessel. We can use it to test drugs before they go to clinical trial,” explained Ding. “This is a profound development and an encouraging first step toward our goal of creating structures that function like a healthy cell should function.”
The researchers are now working on depositing cells during the 3D printing process to make a living artery.
The research paper “Orthogonal programming of heterogeneous micro-mechano-environments and geometries in three-dimensional bio-stereolithography” is co-authored by Hang Yin, Yonghui Ding, Yao Zhai, Wei Tan, and Xiaobo Yin.

For the latest additive manufacturing news subscribe to the 3D Printing Industry newsletter, find us on Facebook and like us on Twitter.
Looking for jobs in engineering? Search open opportunities and apply with 3D Printing Jobs. You can also make a profile to advertise your latest vacancies.
Featured image shows an illustration of arteries. Image via CU Boulder.
Precise Bio, a biotechnology company based in North Carolina, has established a dedicated ophthalmology business unit in its Winston-Salem facility. In addition to its 4D Bio Fabrication Platform, the unit aims to advance 3D bioprinting research for the eyes.
“As the first company to transplant a 3D printed corneal graft in animals, we are uniquely positioned to advance the use of bio-printed tissues in ophthalmology,” ays Aryeh Batt, Co-Founder and CEO of Precise Bio.
“Establishing a business unit dedicated to realizing this potential will support our future financing strategies and ensure that our financial resources are aligned with the tremendous power of our technology and intellectual property.”
Advancing 3D printed corneal grafts
According to Batt, the ophthalmology market has an estimated cumulative value of $10 billion. Precise Bio aims to accelerate the development of autologous cell sourcing, innovative bio-inks, and bioreactors, with an aim to create functional 3D printed organs.
As a result, Precise Bio has developed a laser-assisted 4D bio-fabrication technology platform for cell expansion and the production of complex tissues in a highly reproducible manner. Shay Soker, Co-Founder of Precise Bio and Ph.D., Professor at the Wake Forest Institute of Regenerative Medicine (WFIRM) stated, “[Our] company’s technology overcomes multiple challenges in scalable, reproducible manufacturing of bioprinted tissues and organs, and positions Precise Bio for leadership in the field of regenerative medicine.”
“Precise Bio’s technology has the potential to truly transform the treatment of serious diseases, and to address the unmet needs in corneal replacement and other indications, which cannot be met by the limited number of donor tissues and organs.”
Batt added, “Based in part on the requests and suggestions from key audiences, we are currently pursuing different ophthalmic programs, two of which are being undertaken with collaborators.”
The human cornea program strives to advance the development of other organs and tissues for use in ophthalmic indications. This includes retinal patches, vision correction lenticules (porous outer plant tissue ), and solutions for ocular surface diseases.

3D printing and ophthalmology
Earlier this year, scientists at Newcastle University (NCL) successfully 3D printed human corneas for the first time. Following this, Dr. Song Hongxin, an ophthalmologist and researcher at the Beijing Tongren Hospital, China, 3D printed spectacles to help patients with deformed corneas.
Furthermore, the University of Sydney’s Save Sight Institute was awarded a $1.15 million (AUD) grant to progress commercialization of its 3D corneal biopen, capable of extruding bioink directly onto an eye to aid in the regeneration of cells on corneal ulcers.
For the latest additive manufacturing news subscribe to the 3D Printing Industry newsletter, find us on Facebook and like us on Twitter.
Looking for jobs in engineering? Search open opportunities and apply with 3D Printing Jobs. You can also make a profile to advertise your latest vacancies.
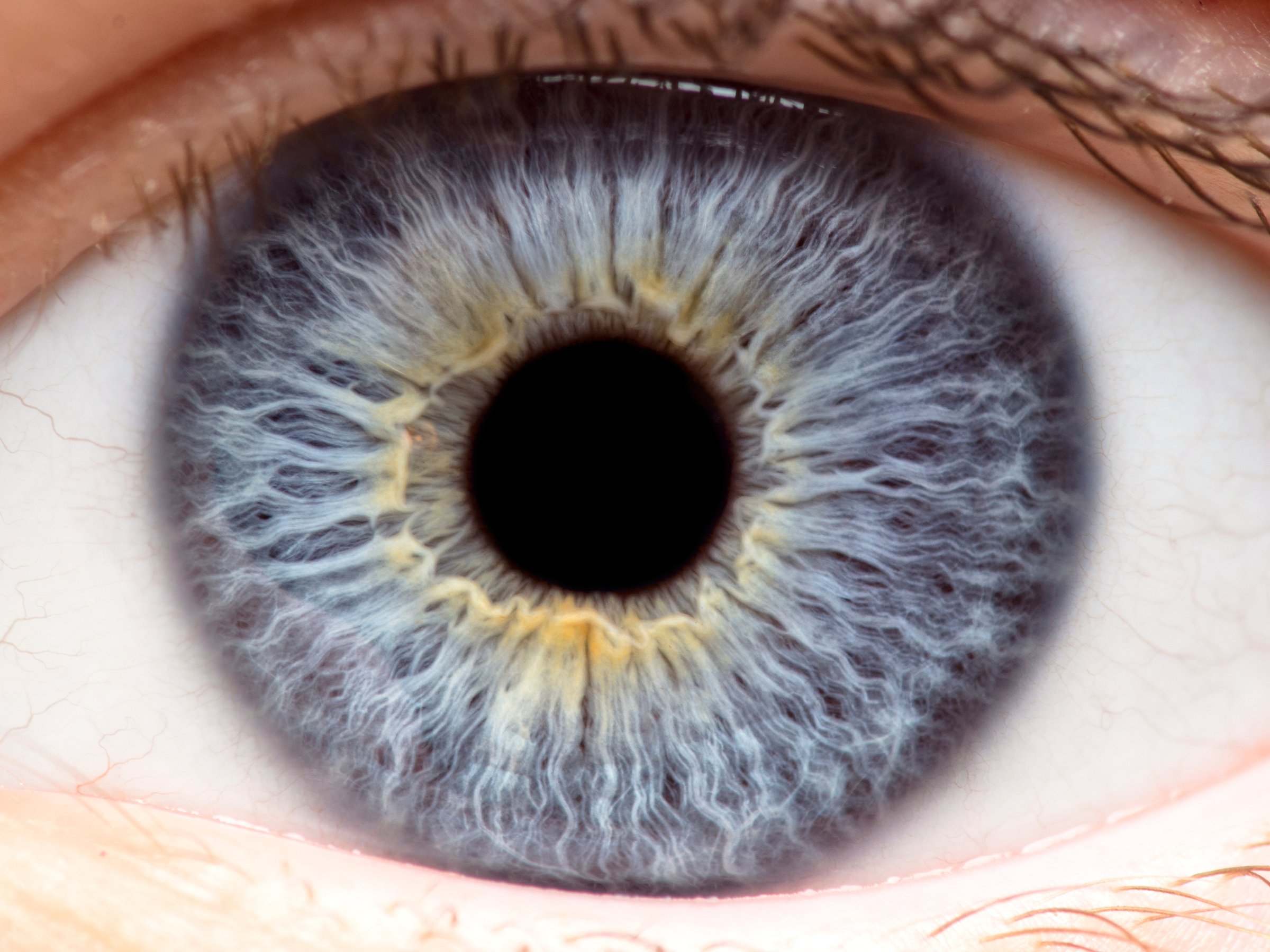
Featured image shows a macro view of the human eye, iris, pupil, eye lashes, eye lids. Photo via Shutterstock.
The Québec Industrial Research Centre (CRIQ), has installed a GE Additive Arcam Electron Beam Melting (EBM) Q10 Plus machine at its facility to accelerate the development of patient-specific lower jawbone implants.
As part of its medical partnership with the University Hospital of Quebec (CHU de Quebec), CRIQ is working with CHU’s cranio-maxillofacial surgeons on applying additive technologies to transform the design to manufacture process of such implants.
“The medical device industry is one of the pioneer industries of additive manufacturing,” said Stephan Zeidler, Business Development, Manager Medical, GE Additive.
“[It] enables companies to manufacture patient-specific implants and customized devices in small batch production, but still in a cost-effective, industrial process. This way, the technology perfectly serves the trend for more individualized treatments in healthcare.”
Quebec invests in additive manufacturing in healthcare
In 2016, CRIQ entered into a medical partnership with CHU – which included five hospitals in its province – to improve medical technologies for better patient care. During this year, CRIQ also partnered with the Canadian Manufacturers and Exporters (CME) to leverage the use of additive manufacturing in Canada.
The following year, CRIQ and the CHU network launched a project to open a $2.874 million (USD) medical 3D printing center in Québec City. This facility will be integrated into all hospitals within the region to provide point-of-care 3D printing services for customized prosthesis as well as research in the field of regenerative medicine.
As a result of its ongoing work with cranio-maxillofacial surgeons, led by François Gingras, Director of Industrial Equipment at CRIQ, the institutions intend to improve the entire design and process cycle as well as validation and medical certification for patient-specific lower jawbone implants. Specifically, the teams are aiming to provide a three-week turnaround of patient-specific implants, rather than the current six weeks required when using traditional manufacturing.
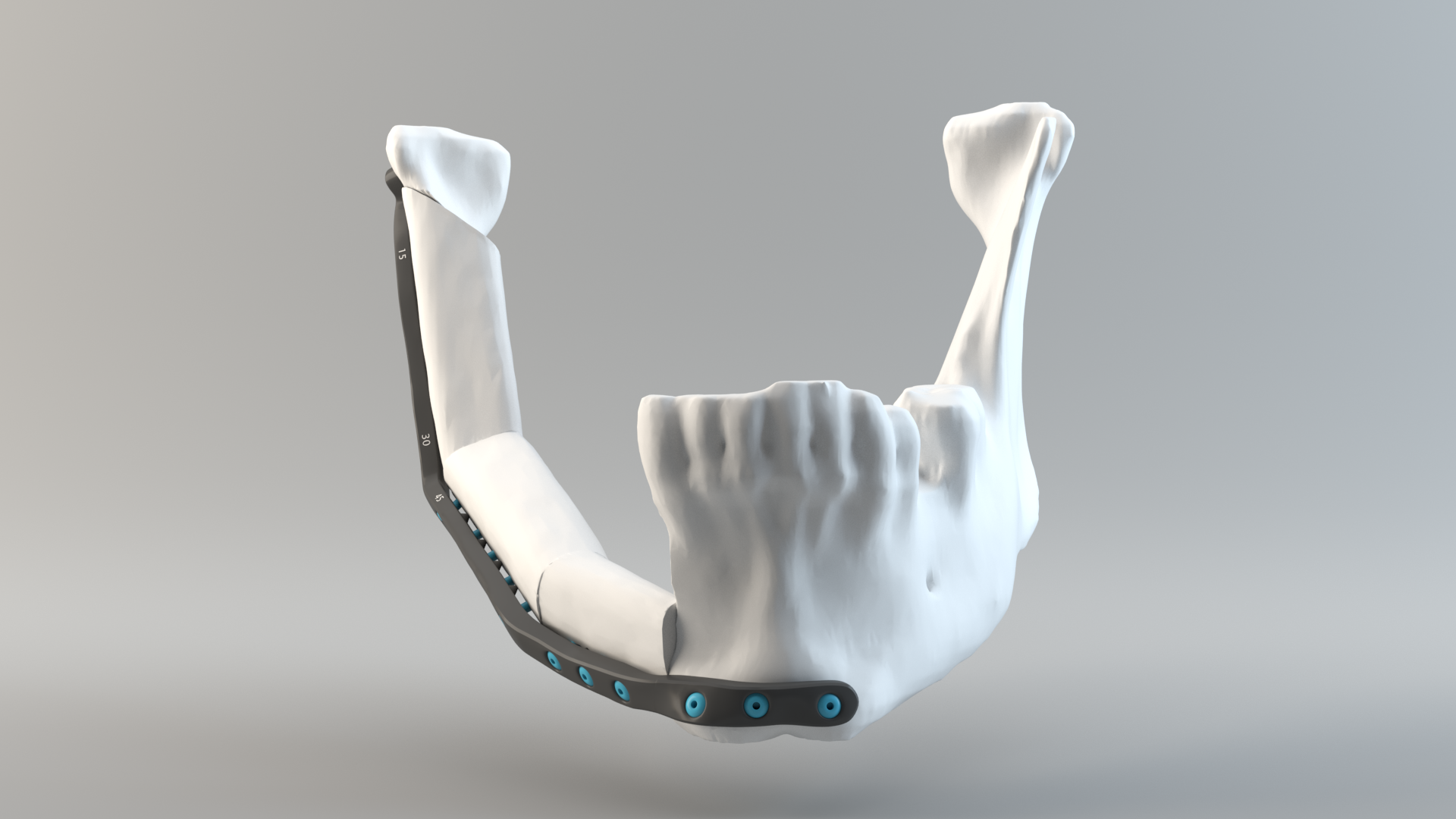
3D printed, patient-specific lower jawbone implants
CRIQ and CHU are tackling the entire supply chain which covers design and CAD of implants, fabrication of the implant, post-processing, cleaning, and sterilization. According to CRIQ, this strategy will define and influence pre-processing, design, testing, and fabrication systems and ensure consistency and repeatability.
Gingras explains that this approach challenges the perception that patient-specific implants are too expensive and not a good business case for additive manufacturing. “CRIQ sees the business case for patient-specific implants at a system level as they cannot be measured on an implant by implant basis.”
“The business case isn’t in a part-to-part comparison; it needs to be justified through system-wide impact. If a patient-specific implant can accelerate patient recovery, reduce risk, and lower overall healthcare costs for the Quebec Government, then we have a business case,” added Gingras.
CRIQ is expected to enter full production of its 3D printed implants by January 2020 upon completion of the process of medical certification of additively manufactured mandibles by Health Canada.

Stay updated with the latest in additive manufacturing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Looking for a change of pace or seeking new talent? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows a 3D model of a lower jaw implant. Photo via Quentin Schneider/ 3D modeling lab, CHU de Quebec.
CollPlant, an Israeli 3D bioprinting company and United Therapeutics Corporation, a Maryland-based biotechnology company, have commenced a license, development and commercialization agreement for the development of 3D bioprinted lung transplants.
Within this agreement, CollPlant will receive $5 million upfront, as well as regulatory and operational milestone payments of up to $15 million to manufacture and supply its bio-ink to United’s organ-manufacturing and transplantation subsidiary, Lung Biotechnology PBC.
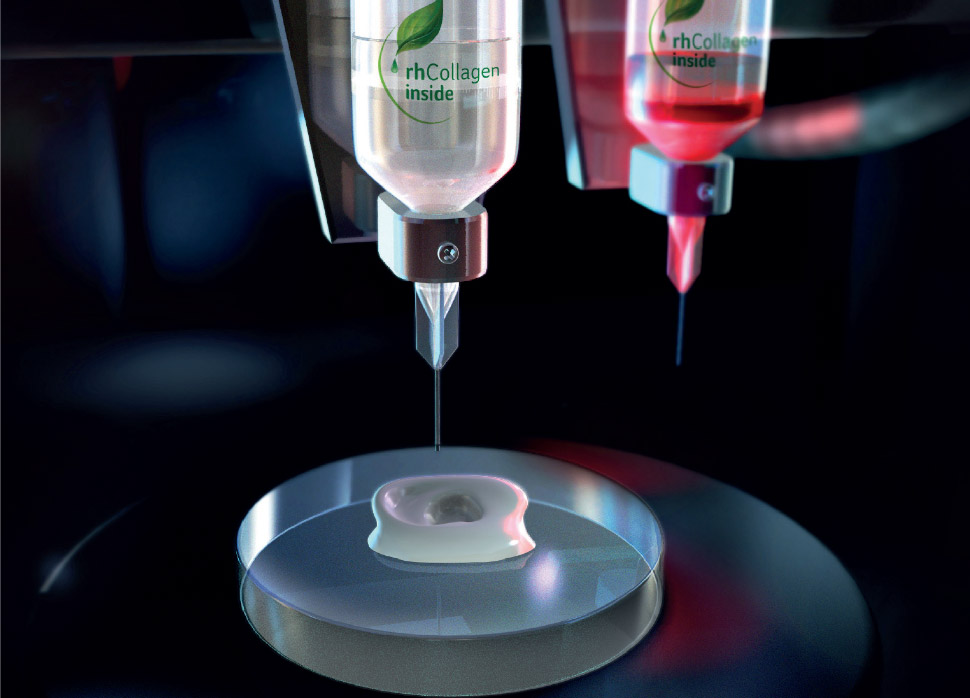
CollPlant’s proprietary recombinant human collagen (rhCollagen) is grown and harvested from tobacco plants engineered with five human genes. This purified collagen is used as a foundation when 3D bioprinting solid-organ scaffolds.

3D bioprinted lung transplants
According to CollPlant, collagen is a primary building block for the human body and has been a vital protein in the medical field as it is essential for tissue repair. Currently, most medical grade collagen derives from cadaveric or bovine (beef) or porcine sources, i.e., animal raw materials. Typically, collagen products are used for bone void fill or soft tissue augmentation procedures, treating chronic wounds, burns, or for cosmetic purposes.
CollPlant’s rhCollagen is produced through genetically engineered tobacco seedlings grown over 8 weeks. The tobacco leaves are then harvested, processed and refined until purified rhCollagen can be extracted. This process also deters disease or pathogen transmission – indirect contact infection, e.g. via needle prick injuries. Martine Rothblatt, Ph.D., Chairman and CEO of United Therapeutics stated:
“CollPlant’s extraordinary Israeli technology to transform the tobacco plant that is so associated with lung disease into a collagen-expressing plant that will be essential to the production of an unlimited number of transplantable lungs.”
Thus, this agreement will utilize rhCollagen and BioInk technology from CollPlant with the regenerative medicine and organ manufacturing experience of United Therapeutics to 3D printed lung transplants for patients in need.
Lung Biotechnology PBC invests in transplantable organ research
Earlier this year, Lung Biotechnology PBC contributed to the $9 million financing awarded to Indiana University School of Medicine (IUSM) to further their 3D bioprinting research for the development of transplantable organs. Prior to this, 3D Systems announced its plans to collaborate with Lung Biotechnology PBC to develop 3D bioprinting solutions.
Stay updated with the latest in additive manufacturing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Looking for a change of pace or seeking new talent? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows an animation showing precollagen fibers inside a single tobacco cell, to be extracted. Image via CollPlant.
3D Systems has announced that Amann Girrbach, an Austrian dental technologies company, will be utilizing its award-winning NextDent 5100 3D printer to optimize dental device production.
As a result, Ceramill, Amann Girrbach’s suite of CAD/CAM software, has been integrated into 3D Systems’ 3D Sprint software for use in the NextDent 5100. This will better equip dental labs and clinics for the faster production of dental devices.
“We are pleased that Amann Girrbach, an industry leader in dental CAD/CAM solutions, is adopting our NextDent 5100 as the additive component in the Ceramill workflow,” said Rik Jacobs, Vice President, General Manager, Dental, 3D Systems.
“This new partnership with Amann Girrbach is an important milestone in our strategy to redefine digital dentistry and demonstrate our ability to optimize production workflows.”

Additive manufacturing dental solutions
With partners in over 90 countries worldwide, Amann Girrbach offers thousands of dental lab customers various solutions using additive manufacturing technologies to produce precision trays, models, surgical guides, dentures, orthodontic splints, crowns, and bridges. From its Ceramill Map 600 high-performance scanner to the Ceramill Therm S speed sintering furnace, Amann Girrbach aims to enable a complete in-house process chain for such digital dental devices.
Using Figure 4 technology, NextDent 5100 is said to have print speeds up to 4x faster and than competitive systems, with significantly reduced manufacturing cost. As an integrated single-source solution, this 3D printer is capable of printing biocompatible dental materials and is targeted towards lab managers, dental technicians, dental prosthetic technicians and clinical prosthodontists, and orthodontists. Christian Ermer, Head of Product Management at Amann Girrbach said:
“With the addition of 3D Systems’ NextDent 5100 3D printing to our Ceramill System, we can offer our customers a more feature-rich digital dental workflow that will make them more competitive and efficient.”
“For example, a dental model is an important part of the restoration process and now with the Next Dent 5100 our customers can print their own models cost-effectively without having to move outside the Ceramill workflow. The NextDent 5100 can also produce bridges, crowns and even the full denture which dramatically lowers production costs and reduces turnaround time. This is certainly a win-win for our customers and their dental patients.”
Catch up with the latest in additive manufacturing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Looking for a change of pace or seeking new talent? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the Ceramill Digital Dental workflow with the NextDent 5100. Image via 3D Systems.
A new 3D bioprinting method has been developed by biomedical engineers from the University of Utah.
With the aim to improve a patient’s recovery, biomedical engineering assistant professor Robby Bowles, and his team, have developed a method to 3D print stem cells from a patient’s own body fat to form tendons and ligaments.
“[This] will allow patients to receive replacement tissues without additional surgeries and without having to harvest tissue from other sites, which has its own source of problems,” said Professor Bowles.

Controlling cell deposition
Following two years of research, Professor Bowles and his team created a 12-channel pilot printhead to enable the deposition of stem cells in complex patterns said to be previously unattainable. The researchers state that “cells that make up a tendon or ligament must gradually shift to bone cells so the tissue can attach to the bone.” Professor Bowles added:
“This is a technique in a very controlled manner to create a pattern and organizations of cells that you couldn’t create with previous technologies. It allows us to very specifically put cells where we want them.”
The bioengineers collaborated with Carterra, Inc., Salt Lake City-based developers of microfluidic medical devices, to create the printhead. This custom part was then attached to a Carterra 3D printer which was formerly used to deposit antibodies for cancer screening applications.
According to the research team, replacement tissue harvested from another part of the patient’s body or a cadaver can be of poor quality. Such areas as spinal discs contain “complicated structures with bony interfaces that must be recreated to be successfully transplanted.” This motivated the team to create a new 3D printing technique to remedy such problems.
Furthermore, Bowles believes the technology in the printhead could be adapted for any kind of 3D printer and contributes to the goal of 3D printing whole organs.
Regenerative medicine and 3D printing
Additive manufacturing has proven to be an asset in biological studies and experimentation. In the realm of regenerative medicine, 3D bioprinting has allowed for the creation of cell structures that work to heal wounds inside the human body.
Earlier this year, researchers from George Washington University (GWU) developed a 3D bioprinting technique to create multi-responsive smart structures for nerve regeneration. Prior to this, a study produced by four research institutions in Brazil demonstrated the ability of two 3D printable biocompatible polymers to create structures ideal for cell-growth.
The method article “Microfluidic Flow Cell Array for Controlled Cell Deposition in Engineered Musculoskeletal Tissue” is co-authored by David Ede, Nikki Davidoff, Alejandro Blitch, Niloofar Farhang, and Robby D. Bowles.
For more on the latest additive manufacturing news, subscribe to the 3D Printing Industry newsletter, like us Facebook and follow us on Twitter.
Looking for a change of pace or searching for talent? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows University of Utah biomedical engineering assistant professor Robby Bowles with a customized 3D bioprinter. Photo via Dan Hixson/University of Utah College of Engineering.
Poietis, a French biotechnology company, has announced a partnership with Servier Laboratories, a privately owned global pharmaceutical company, to use 3D bioprinting technologies to create a 4D liver toxicity detection model capable of identifying drug-induced lesions.
“We believe 3D Bioprinters are more than robotic pipettes,” Bruno Brisson, Co-Founder and Vice President of Business Development at Poietis, told 3D Printing Industry.
“So, the 4D Bioprinting paradigm is: guiding or programming self-organization. The goal is then to determine appropriate 3D micro-patterns (blueprints) of tissue components (cells) so that specific tissue function emerges with time considering both internal interactions and interactions with host (external).”
Combating drug-induced liver disease
Drug-induced liver disease occurs as a result of medications, such as vitamins, hormones, herbs, as well as drugs, and environmental pollution. This toxicity in the liver causes lesions, which if not detected, can lead to liver failure and hepatotoxicity – chemical-driven liver damage.
The scientific partners have identified weaknesses in current preclinical detection models which use animal models and cell cultures of human liver cells to predict toxicity. As these models cannot fully replicate the human liver, the results are limited as the hepatotoxic potential is poorly detected.
Liver toxicity testing is significant during preclinical trials with investigational drugs, thus, the scientists have developed a bioprinting method which uses different types of human liver cells and immune cells that imitates the complex multicellular tissue structure of the liver. This model will demonstrate higher accuracy than its predecessors, therefore becoming a vital tool when testing for adverse effects of developing medicinal products.
“We actually introduced the concept of 4D Bioprinting as a new paradigm for engineering complex tissues with our laser-assisted bioprinting technology for more than 3 years,” explained Brisson.
4D printing in medicine
Brisson explains that standard in vitro liver models used in drug analysis is produced with a two-dimensional single layer cell tissue. However, with advancements in 3D printing for regenerative medicine, scientists have been able to create 3D printed cell scaffolds for cell growth.
As the cell structures grow, they can be used to repair parts of the human body, particularly organs, with the body’s original cells. Combined with the concept of 4D printing,3D printed structures with morphing capabilities, the cell structures are able to expand as a result of its changing environment i.e. water and heat. Brisson added:
“[Time] is something that needs to be taken into account since the conception of your bioprinting processes for a given specific tissue (and especially with biological and cytocentric CAD software).”
The model will be validated by testing a panel of compounds known to induce, or not induce, liver toxicity. The liver model product is expected to be ready in approximately 18 months.
Keep up with the latest 3D printing news by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Searching for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows Poietis’ Cytocentric CAD tool which can define and locate the position and environment of each cell within three-dimensional tissue structures. Image via Poietis.
Scientists from the University of Toronto (UoT) and Sunnybrook Research Institute (SRI), have created a handheld 3D bioprinter capable of printing skin cells to treat deep-thickness wound on burn victims.
According to the research recently published in the Lab on a Chip journal, “When manually positioned above a target surface, the compact instrument [weighing 0.8 kg] conformally deposits a biomaterial or tissue sheet from a microfluidic cartridge.”

The ‘first’ handheld bioprinter
According to the World Health Organization (WHO), an estimated 180,000 deaths annually are caused by burns – the vast majority occur in low-and-middle-income countries. Seeking methods to treat burn victims remotely, Navid Hakimi Ph.D. student at UoT, and Dr. Marc G. Jeschke, a Professor of Immunology at UoT and Director of the Ross Tilley Burn Centre at Sunnybrook Hospital led the research of the portable 3D bioprinter.
“Most current 3D bioprinters are bulky, work at low speeds, are expensive and are incompatible with clinical application,” said Dr. Axel Guenther, Associate Professor at UoT’s Faculty of Applied Science & Engineering, who supervised the research project.
Bioprinting versus skin grafting
Skin grafting, the surgical transplantation of skin tissue, requires a significant amount of healthy donor skin to travel across the three layers of the skin on deep wounds – the epidermis, dermis, and hypodermis. Nevertheless, according to the research, large amounts of healthy donor skin is not always available.
With minimal operating training, the handheld bioprinter deposits epidermal and dermal cells as well as a mixture of bio-neutral polymers and proteins for even distribution of the cells layers. This process has the potential to replace skin grafting processes as the “skin” printed can be layered in multiple sessions to heal a deep-thickness wound.
The scientists have also incorporated hyaluronate, a protein found in human connective tissue into the printer model. This protein has the ability to promote healing and integrity among cells and collagen. A test was conducted on agarose (a linear polymer) substrates in in vitro conditions. This process validated the bioink’s ability to be extruded inconsistent narrow sheets with the desired content, width, and thickness.
Following these tests, the team conducted in vivo studies, which applied the entire 3D printer to deep skin wounds in experimental pig and mouse skin. The bioink stripes were able to cover the wounds in their entirety, using various angles. The technology is now being developed for human testing.

3D printing and wound care
Additive manufacturing has contributed to innovative solutions for medical skin treatments and wound care. Last year, scientists from the VTT Technical Centre of Finland, a state-owned research and development non-profit, have developed a nanostructured cellulose 3D printable smart-dressing and 3D printed circuit board that heals and monitors skin wounds.
Prior to this, a research team at Pohang University of Science and Technology (POSTECH) in Korea, developed a skin 3D bioprinting process which uses extrusion-based and inkjet printing depositing advanced bioink materials. This method accelerated the growth of tissues in the layers of human skin, demonstrating a substantial skin culture in 2 weeks.
The research paper, “Handheld skin printer: in situ formation of planar biomaterials and tissues” was co-authored by Navid Hakimi, Richard Cheng, Lian Leng, Mohammad Sotoudehfar, Phoenix Qing Ba, Nazihah Bakhtyar, Saeid Amini-Nik, Dr. Marc G. Jeschke, and Axel Guenther.
Stay updated with the latest scientific developments in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Searching for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the handheld 3D bioprinter depositing skin cells onto a substrate.Clip via UoT.