Johnson & Johnson Medical GmbH, the subsidiary of Johnson & Johnson Medical Devices Companies (JJMDC) has acquired spinal implant 3D printing specialist Emerging Implant Technologies (EIT), headquartered in Germany.
Following this acquisition, Johnson & Johnson’s orthopedics branch DePuy Synthes will also strengthen its interbody implant portfolio which includes titanium integrated PEEK technology for minimally invasive spinal surgery.
“Our goal is to offer a complete portfolio of interbody solutions that provides surgeons with even more options for the treatment of their patients,” said Aldo Denti, Company Group Chairman of DePuy Synthes. The financial terms of the transaction remain undisclosed.

3D printing at Johnson & Johnson
With over 130 years of experience, Johnson & Johnson is a globally recognizable healthcare company, operating across 57 countries. The company’s overall mission to “profoundly change the trajectory of health for humanity” has prompted various ventures into new technologies such as additive manufacturing.
Most recently, the company collaborated with Trinity College Dublin (TCD), and the AMBER research center to establish a new specialist 3D bioprinting facility for developing personalized healthcare solutions. Prior to this, Johnson & Johnson partnered with Aspect Biosystems to leverage 3D bioprinting to explore treatment common knee injuries. The company has also partnered with Carbon and HP in order to create custom, 3D printed surgical devices.
Through a previous deal between Tissue Regeneration Systems and Materialise, Johnson & Johnson subsidiary DePuy Synthes offers the 3D printed titanium TRUMATCH maxillofacial implants for jaw and facial reconstructions, and 3D printed bioresorbable patient-specific implants. Now, with a focus on treating spinal diseases, DePuy Synthes has assimilated EIT’s proprietary advanced cellular titanium 3D printing technology into its portfolio.
Medical 3D printing market consolidation
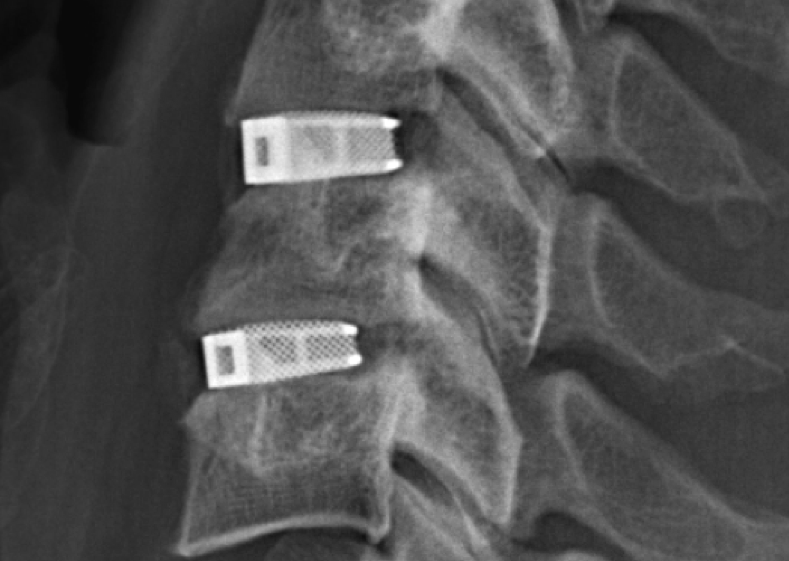
EIT Cellular Titanium implants are made using Selective Laser Melting (SLM), forming open and interconnected porous structures designed to promote bone in-growth. Last year, EIT’s Cellular Titanium implants received 510(k) clearance from the FDA. Prior to this, the implants had been used in 10,000 cases in over 15 countries around the world.
In recent years, there has been a proliferation of 3D printed spinal implants achieving clearance for the commercial market. Now, it seems, the market is experiencing some consolidation. Johnson & Johnson’s EIT acquisition comes just after FORTUNE 500 medical technology firm Stryker acquired Virginia medical device manufacturer K2M in a $1.4 billion deal.
“We are excited to welcome the skilled team at EIT,” concludes DePuy Synthes chairman Denti, “and together, we aspire to bring to market technologies that allow surgeons to perform spinal fusion procedures reliably and with consistent outcomes.”
Keep up with the latest 3D printing news by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Searching for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the surface structure of an FDA approved 3D printed cellular titanium implant. Image via EIT.
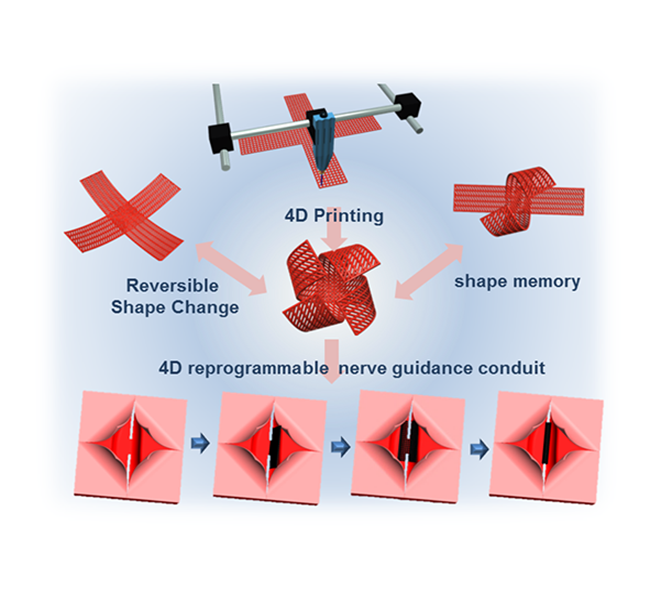
Researchers from George Washington University (GWU) have developed a 4D bioprinting technique to create multi-responsive smart structures for nerve regeneration. In a proof-of-concept study published in Advanced Biosystems, the researchers produced a smart nerve guidance conduit capable of “blooming like a flower.”
Blossoming 4D printed biostructures
Regenerative medicine research working towards functional artificial organs has enabled 3D printed complex vascular tissue structures and cell scaffolds for repairing the damage to the human body.
Nevertheless, according to the study, 3D printed biostructures with integrated time-dependent shape changes, i.e. 4D printing, “offers the opportunity to realize more complicated folded structures in biomedical devices or soft robotics, as well as in tissue engineering to mimic dynamic changes of native tissues and organs.”
The GWU researchers propose a stereolithographic 3D printing method applied with the “universal concept of stress‐induced shape transformation” to achieve the 4D dynamic reversible movement reminiscent of a flower in bloom. The following clip from GWU demonstrates the blooming movement.
SLA 4D printing
Using a naturally derived photocrosslinkable monomer – Soybean Oil Epoxidized Acrylate (SOEA) – as an ink, the GWU team were able to create a solidified flat star structure as a result to UV light‐induced graded internal stress.
The SOEA is then washed with ethanol for solvent‐induced relaxation which dynamically transforms the structure into a claw formation. This change is autonomous and reversible. Furthermore, various nanoparticles, such as nanohybrids with graphene, are incorporated into the structures during the printing process for further shape modification. This addition has been used to generate structures resembling flying birds.
Following an in-depth analysis, the researchers concluded that these smart structures can provide “outstanding multifunctional characteristics for nerve regeneration including physical guidance, chemical cues, dynamic self‐entubulation, and seamless integration.”
“By employing this fabrication technique, creating multiresponsive smart architectures, as well as demonstrating application potential, this work paves the way for true initiation of 4D printing in various high‐value research fields.”
The research paper “Stereolithographic 4D Bioprinting of Multiresponsive Architectures for Neural Engineering“ is co-authored by Lijie Grace Zhang, Shida Miao, Haitao Cui, Margaret Nowicki, Lang Xia, Xuan Zhou, Se‐Jun Lee, Wei Zhu, Kausik Sarkar, and Zhiyong Zhang.
For more of the latest research related to 3D printing sign up to the 3D Printing Industry newsletter, Also, follow us on Twitter, and like us on Facebook.
On the lookout for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows blossoming 4D smart structures forming birds and flowers. Photo via GWU.
HP has announced its participation in the Centers for Disease Control and Prevention (CDC) pilot program that aims to develop new antibiotics designed to fight antimicrobial-resistant bacteria.
According to the CDC, a U.S. healthcare organization, infectious diseases with resistant organisms caused by antibiotics are difficult to treat, requiring costly and sometimes toxic alternatives. With HP’s bioprinting technology, microbiologists are able to print antibiotics samples for testing directed at halting the spread of such bacteria.
“Bacteria continuously develop new ways to resist antibiotics—once a drug is approved for use, the countdown begins until resistance emerges,” said Jean Patel, PH.D. D (ABMM), Science Team Lead, Antibiotic Resistance Coordination and Strategy Unit at CDC.

The HP D300e Digital Dispenser BioPrinter
The ability of a microbe to resist the effects of medication, i.e., antimicrobial resistance, remains an international public health issue as it opens the door for a plethora of diseases to spread, states the CDC. Without proper testing, new drugs used to combat such bacteria can contribute to the presence of antimicrobial resistance.
Thus, with the use of an HP D300e Digital Dispenser BioPrinter researchers can properly analyze the behavior of antimicrobial bacteria for effective antibiotics. Similar to the set-up of a regular ink printer, the HP Bioprinter dispenses volumes from picoliters to microliters of small molecules for drug discovery, and research into microbiological structures(genomics and proteomics).
In an effort to develop new antibiotics, the CDC is also planning to deploy HP BioPrinters within its Antibiotic Resistance (AR) Lab Network across four regional areas in U.S. Additionally, California-based pharmaceutical company Gilead, is using the HP BioPrinter to develop tests for drugs used against the Ebola virus.
“To save lives and protect people, it is vital to make technology accessible to hospital labs nationwide. We hope this pilot will help ensure our newest drugs last longer and put gold-standard lab results in healthcare providers’ hands faster,” added Patel.
As of yet, there is no news on whether HP is developing a 3D bioprinter.
Combating diseases with 3D printing
Earlier this month, researchers from McMaster University in Ontario, presented a 3D printed solution for combating superbugs – a strain of bacteria immune to antibiotics. The Printed Fluorescence Imaging Box (PFI Box), assembled from six 3D printed walls, acts as an experimental chamber for bacteria.
LED lights within the box are used to excite the bacteria’s fluorescent proteins, then a Raspberry Pi processor records the data from this reaction to be analyzed for an appropriate and innovative drug response.
Prior to this, researchers from Northeastern University, inspired by the naturally repellent surface of cicada insect wings, began using Oxford Performance Materials’ (OPM) 3D printed PEKK plastic to develop anti-infection orthopedic devices.
For more of the latest 3D printing news subscribe to the 3D Printing Industry newsletter and follow us on Twitter, and Facebook.
Pursuing a career in additive manufacturing or looking for skilled individuals in the field? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the HP D300e Digital Dispenser BioPrinter. Photo via HP.
Shalini Guleria, Masters student at the University of Waikato, New Zealand, is aiming to 3D print breast tumor cells to identify the best treatment for cancer patients.
With the intentions of reducing long-term, painful cancer treatments such as chemotherapy and radiotherapy, Guleria plans to grow and test cancer tumors outside of a patient’s body using real cells. These bioprinted tumors will follow the design of her 3D printed plastic tumor models.
“In the future what could happen is if someone has breast cancer, we could take their tumor cells and print out a tumor and try out different drugs on it and see which treatments work and what works best for the patient, explained Guleria. “It’s all about making treatment more patient-specific.”

Growing a 3D printed tumor
To support her research, Guleria has printed webbed, palm-sized hemispheres – resembling breast cancer tumors – which will be the basis for her bioprinted models. Guleria will replace the plastic from the prototype model with cancer cells (known as MCF-7), hydrogel and additional binding materials, which will ultimately grow into a life-sized tumor.
As the bioprinted tumor mesh grows, Guleria plans to observe the rate at which it increases in a 3D model when compared with a 2D model. “Currently the easiest way to do pharmaceutical tests on cancer cells is using 2D models which is basically a petri dish where the cells stick to the bottom of the dish and you analyze them. But humans are three dimensional and 2D doesn’t really present an actual human tumor,” said Guleria.
“Once the tumour is printed we will be able to slice it and look into the depths of the cell. You can look at how the tissues are growing, the fibres connecting and the cell organelles – the things that make the cell grow – how they differ to the 2D model.”
Guleria also plans on using Cisplatin, a common breast cancer chemotherapy drug, on the fully-grown bioprinted tumors to observe its reactions. This experimentation could potentially decrease the dosages, treatment time, and trauma experienced by a real cancer patient.

Attacking cancer with bioprinting
Guleria, who is studying for a Master of Science with a focus on tissue engineering, became interested in cancer research after the death of a close friend who suffered from leukemia.
“She was only 17 and passed away a year after her diagnosis. I was always interested in medical science but when that happened it really hit me and to grieve I went crazy into cancer research,” said Guleria.
Giving cells a 3D environment in order to analyze disease progression is just the first step for Guleria’s research. Her long-term goal seeks to use patient-specific cells to create to determine the most effective cancer treatments.
“We may be able to take the cells from someone who has cancer, and use them to produce something we can test very specifically for that patient,” added Guleria.
“Then we could make a detailed treatment plan for an individual’s body, rather than take a generic approach. A panadol might work for you, and not someone else – everyone’s bodies are different.”
Guleria’s research is currently supervised by Dr. Linda Peters, Senior Lecturer of Biological Sciences at the University of Waikato. Furthermore, Andrew Howard, a Master’s of Science (Research) student, is contributing to this project.
Additive manufacturing and cancer research
From 3D printed breast cancer detectors to patient-specific 3D printed molds for breast reconstruction, additive manufacturing has created innovative and advanced methods to improve the treatment of women with breast cancer.
Moreover, bioprinters have enabled more detailed and accurate research for observing cancer cell behavior. Last year, researchers from the Indian Institute of Technology Hyderabad stated that the benefits of bioprinting include: “the ability to precisely place cells and reproduce in vivo tumor microenvironments, enable high throughput screening of drugs and aid the development of next-generation molecular therapies.”
Keep up with the latest news 3D printing sign up to the 3D Printing Industry newsletter, Also, follow us on Twitter, and like us on Facebook.
Are you searching for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the 3D printed plastic tumor models. Photo via the University of Waikato.
The University of Sydney’s Save Sight Institute has been awarded a $1.15 million (AUD) grant from the New South Wales (NSW) Government’s Medical Devices Fund (MDF) to progress commercialization of its 3D corneal biopen.
The iFix Pen, a hand-held co-axial 3D printer, extrudes bioink directly onto an eye to aid in the regeneration of cells on corneal ulcers. The pen also creates a biological barrier towards ongoing cornea damage caused by infections.
Brad Hazzard, the New South Wales Minister for Health and Medical Research, announced the recipients of the MDF, last Wednesday at the Parliament House in Sydney, Australia.
“Since the Medical Devices Fund began in 2013, the NSW Government has awarded more than $50 million in grants to 31 technologies,” said Hazzard.
“We are committed to supporting innovators in the medtech industry in getting their brilliant ideas off the ground and ultimately saving millions of lives around the world.”

The iFix Pen project
The iFix Pen Project is led by Gerard Sutton, ophthalmologist and Professor of Corneal and Refractive Surgery at the Sydney Eye Hospital, University of Sydney and Professor Gordon Wallace, Founder and Director of the Intelligent Polymer Research Institute, University of Wollongong (UOW).
According to the Save Sight Institute, corneal ulceration, a painful open sore on the clear front surface of the eye which can lead to blindness, is a common problem in Australia and developing countries. Approximately 55,000 Australians with corneal ulceration present themselves to hospitals for treatment each year.
Thus, Professor Sutton and Professor Wallace began developing the iFix Pen Project as an alternative treatment that will potentially reduce the number of patients afflicted with corneal ulceration. The team’s research, which was first reported in the journal Biofabrication in 2016, has shown that the iFix Pen can accelerate feeling, minimize pain and reduce patient recovery time, through the deployment of antibiotics.
The project also collaborates with Professor Peter Choong, Sir Hugh Devine Chair of Surgery, and Head of Department of Surgery at the University of Melbourne, and the NSW Organ and Tissue Donation Service.

The Big Idea Grant
In 2017, the iFix Pen project was awarded $45,000 (AUD) in pre-seed funding through Sydney Local Health District’s Big Idea grant which the research team used to commence surgical tests on sheep.
“Winning the Big Idea last year gave us two things,” Professor Sutton says. “It gave us credibility when we were applying for other grants and the self-belief that we had a great idea worth developing.
“The iFix pen is one part of an overall corneal bioengineering project and, with the support from The Big Idea and the Medical Devices Fund, we are also hoping that within the next five to 10 years, we will be able to develop a 3D bioengineered cornea.
Last year, Professor Wallace’s UOW research team was also awarded a $347,070 (AUD) LIEF grant for a 3D Additive Bio-Fabrication Facility to create the next generation of bioprinting methodologies and 3D fabrication tools.
The iFix Pen is expected to undertake a phase 1 human trial in 2019.

Stay abreast of the latest news in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
On the lookout for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows Dr. Stephen Beirne of the Australian National Fabrication Facility with the iFix Pen. Photo via Australian Research Council Centre of Excellence for Electromaterials Science (ACES).
Dr. Michael Laufer, Founder and Chief Spokesman of the Four Thieves Vinegar Collective, an anarchist biohacking network, debuted a 3D printable kit for homebrewing medication at the Hackers On Planet Earth (HOPE) Conference in New York City, last month.
Acting as an accessible alternative to industrial chemical reactors, this Apothecary Mircolab is capable of synthesizing homemade medication for HIV, opiate overdoses, and pharmaceutical abortions.
This chemical reactor “can synthesize household poisons into literally a life-saving drug,” said Dr. Laufer in a recent interview.

A 3D printed chemical reactor for “homemade free drugs”
Founded in 2015, the Four Thieves Vinegar Collective began clandestine chemistry (chemistry carried out in secret) to “defend people’s right to attempt their own medical treatment.” With this concept, the Collective has developed and published instructions for a 3D printed Epipen, i.e, the EpiPencil, and the Apothecary MicroLab, an automated DIY device designed to make a variety of medications.
During the 11th HOPE conference in 2016, the Four Thieves Vinegar Collection went public and debuted the first generation of the Apothecary Microlab and EpiPencil. Since then, its DIY chemical reactor has developed, and been involved in hacking medical hardware and more complex drug production processes.
The new and improved Apothecary Microlab consists of a small mason jar mounted inside a larger mason jar with a 3D printed lid. Furthermore, the kit contains a 3D printed stepper motor, syringe pump, coupler and shredded shaft which are connected using small plastic hoses.
A thermistor is then attached through the lid to circulate fluids to induce the chemical reactions necessary to manufacture various medicines. The whole process is automated using a computer.
According to the Collective, the 3D printed chemical reactor kit has successfully produced Naloxone (aka Narcan), a drug for opiate overdoses, Cabotegravir and Daraprim, drugs used to treat infections in people with HIV, and Mifepristone (aka RU486), and misoprostol, two chemicals needed for pharmaceutical abortions.

Open-source medical 3D printing solutions
Additive manufacturing enables a cost-effective alternative to traditionally expensive laboratory equipment. Whether it is used amongst biomedical researchers, or more recently, the general public (as a result of groups such as the Four Thieves Vinegar Collective), the presence of 3D printed customized labware and reaction vessels has increased.
Recently, researchers from Imperial College London (ICL) created an inexpensive open-source 3D printable membrane feeder to facilitate a wider range of laboratories with customizable tools for scientific malaria experimentation.
Prior to this, researchers from New York Genome Center and New York University developed the open-source 3D printed microfluidics control instrument for cell analysis which is 20 to 200 times cheaper than its traditional counterpart.
In addition, new research published in Additive Manufacturing, outlines the potential of 3D printed customizable toolkits and reaction vessels for chemically resistant lab equipment using affordable material and FFF/FDM 3D printers.
Stay abreast of the latest news in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
On the lookout for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows Dr. Michael Laufer with the 3D printed chemical reactor kit at the HOPE conference. Image via Michael Laufer/Youtube.
The Mayo Clinic, a non-profit academic medical center based in Rochester, Minnesota, implemented additive manufacturing into its pre-surgical planning processes over ten years ago.
Now, with several successful cases, Dr. Jonathan Morris, Co-Director of the Anatomical Modeling Laboratory and Neuroradiologist at the Mayo Clinic, reflected on the positive impact 3D printed anatomical models have had in addressing unique and complex surgeries.
“3D Printing bridges this gap of taking the unknown and making it known,” said Dr. Morris in a keynote speech at the Materialise World Summit, in Brussels, Belgium.
“The trouble with just having a three-dimensional object on a two-dimensional screen is that it’s still two dimensional. So, if you’re presenting the skull of a two-year-old and an 82-year-old there the same size on the screen – you don’t have any sense of scale.”
Mayo’s Anatomical Modeling Lab
Founded over 150 years ago, the Mayo Clinic, which maintains three campuses across the U.S., treats an estimated 1.3 million patients from 135 countries each year. Within its annual patient treatments, approximately 76,000 surgeries are performed.
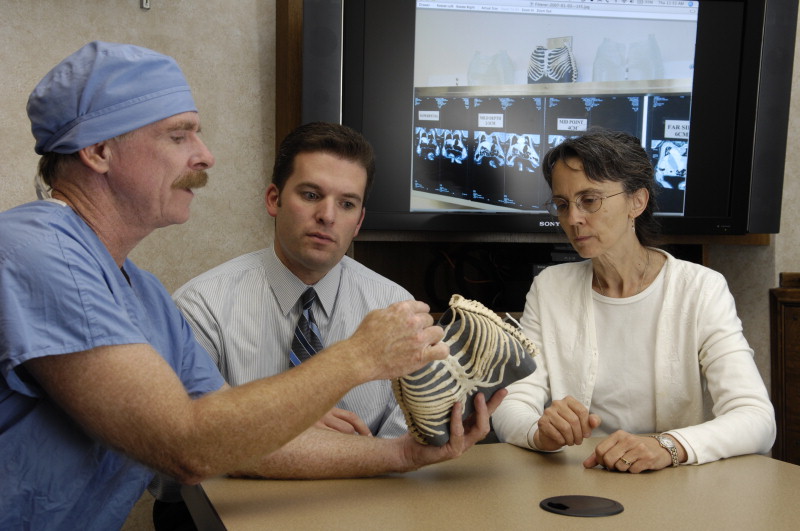
Among the vast amount of surgical cases, in 2008, Dr. Christopher Moir, Pediatric Surgeon at the Mayo Clinic, was confronted with five-month-old conjoined twins that were fused at the chest.
Considering the unusual nature of this case, Dr. Moir and his surgical team sought out Dr. Morris, who had previous 3D printing experience from his time at the National Institutes of Health (NIH), to create customized anatomical models of the twins’ shared organs and rib cage.
These models aided the pediatric team in finding the correct passages and methods to safely separate the twins. In 2016, the widely publicized case of the formerly conjoined Mcdonald twins introduced the capabilities of 3D printed medical models to optimize surgical planning for complex operations. Dr. Morris stated:
“You get an X-ray, CT scan, or an MRI which provides information but until you print that object out and have it in your hands in life-size you’re still in a state of relative uncertainty.”
Following the successful surgical separation of the five-month-old conjoined twins and an influx of similar cases, Dr. Morris and Dr. Jane Matsumoto, Radiologist at the Mayo Clinic, established the Anatomical Modeling Lab.
In-hospital 3D printing
Offering clinical 3D printing services to its three U.S. campuses was not an easy task, explained Dr. Morris. Thus, members of the lab set up a validation workflow, including an ordering system, CAD software (i.e the Materialise Mimics Innovation Suite), and 3D printing systems within Mayo Clinic Operating Rooms (OR).
“[3D printing] has to happen where the doctors are. The fact that it’s in-house makes the lab very accessible for everyone [meaning] surgeons can simply come by between surgeries, explain what they want to visualize, and leave the rest to the radiologists.”
Over the past few years, companies and medical facilities have been integrating additive manufacturing technologies for 3D printing labs within hospitals. Stratasys began installing its Mojo Desktop 3D Printer in five Veterans Affairs (VA) hospitals throughout the U.S. Following this, 3D LifePrints raised £500,000 in Series A funding to expand its medical 3D printing manufacturing operations international level.
With such developments, experienced surgeons and emerging medical practitioners can use 3D printed anatomical models as an education tool within unique and complicated surgical procedures.
Dr. Morris explained,“Surgeons really want to be able to shorten the gap of their trainees to get from knowledge to experience. And 3D Printing does that.”

Catch up with the latest news in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Seeking a new position in the additive manufacturing industry? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows Dr. Jonathan Morris giving his keynote presentation at the Materialise World Summit, in Brussels, Belgium. Photo via Materialise.
Researchers from the University of Toronto (UOT) have developed an open-source, low-cost 3D bioprinter for constructing complex vascular tissue structures.
With this custom bioprinter, the UOT researchers conducted a study seeking to resolve a concern within tissue engineering which derives from the poor development of vasculature – the arrangement of blood vessels in an organ or body part.
Using sacrificial materials such as gelatin and pluronic F-127 (PF127), a thermogel polymer, the research determined PF127 as the superior material for use in developing vascularized tissues. According to the research paper, this discovery “can potentially help accelerate the widespread adoption [of] 3D bioprinting to create such engineered tissues.”
This research contributes to the ambitious goal of many scientists working in regenerative medicine – which is to create functional 3D printed organs.

Branching vessel channels with bioprinting
Vascular channels are extremely important with the human anatomy as they encompass the system of blood vessels that supply blood through tissues to our organs, and ultimately our entire body.
These intricate channels are challenging to replicate using current tissue engineering methods, however, 3D bioprinting has enabled the creation of complex structures of varying lengths that can branch out and interconnect within a single construct.
Moreover, hydrogels, a polymeric biomaterial, have played an integral part in this research as their hydrophilic capabilities enable them to hold large amounts of water in a 3D networks. This permits cell growth and tissue organization, which leads to the formation of vascular channels.
This class of biomaterials has been used frequently within 3D bioprinting experimentation due to its structural similarity to a cell’s natural extracellular matrix (ECM).
Experimenting with gelatin and PF127
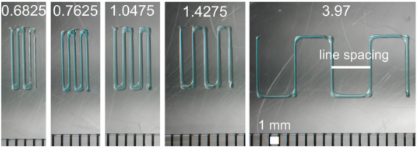
During this experiment, UOT researchers used their custom 3D bioprinter, which cost an estimated $3,000, to print the gelatin and PF127 materials within hydrogel constructs into a desired vascular pattern.
The modular 3D bioprinter included extruding systems 3D printed from ABS, to hold sterile 10 ml syringes. In addition, the printing system operated from an open-source Duet v0.6 controller board, which to managed five-axis motors (one in X, one in Y, and three in Z) and five independent extruders.
With a sufficient print resolution of 0.5–1 mm, the bioprinter successfully processed the sacrificial materials, maintaining a uniform filament diameter, to replicate complex vascular tissue structures. The materials were then compared based on its consistency and accuracy.
The study concludes, “We found Pluronic F-127 to produce filaments with higher spatial resolution, greater uniformity, and greater elastic modulus than gelatin filaments, and with low toxicity despite being a surfactant, making it particularly suitable for engineering smaller vascular conduits (channels).”

Stay abreast of the latest news in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
On the lookout for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows UOT custom bioprinter.
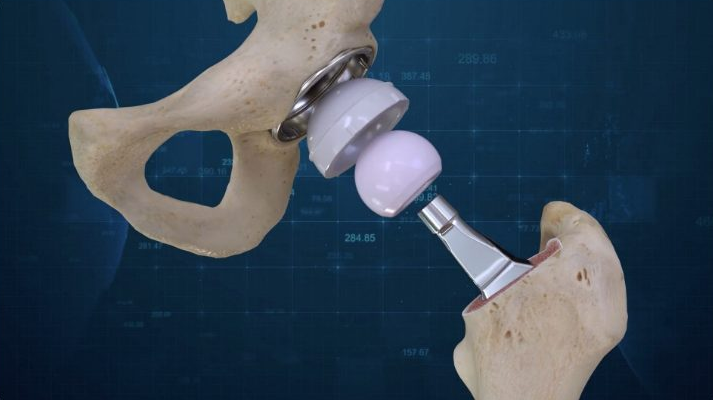
Last week, ConforMIS, a medical technology company based in Massachusetts, launched its first two 3D printed hip system implants which, according to ConforMIS, is “the first ever 3D designed primary total hip replacement system.”
A day prior to this launch, on July 31st, the first surgeries using this system were performed on by Gregory Martin, M.D., Joint Fellowship Trained Orthopedic Surgeon at the JFK Medical Center in Atlantis, Florida.
“The ConforMIS Hip System is designed to address many of the shortcomings of primary hip replacement today. For the first time, orthopedic surgeons have a fully-guided system designed to address the wide variations in anatomy presented across our cases,” said Dr. Gregory Martin M.D, a Surgical Consultant within Conformis’ design team.
Adapting to new medical technologies
Within the past two years, the FDA has cleared a number of 3D printed medical implants to be surgically placed within the human body. Considering the emergence of approved 3D printed implants as well as 3D printed surgical devices, surgeons must learn and adapt to these new medical technological solutions.
Hip implants, in particular, have a significant function within the human body. This is because they act as a replacement for diseased or damaged areas of the hip joint – which is responsible for a wide range of body movement. And, with additive manufacturing, implants can be created to perfectly match for a patient’s anatomy.
According to Mark Augusti, CEO and President of ConforMIS, the hip replacement global market is worth an estimated $7 billion. Thus, with the release of the ConforMIS Hip System Augusti expects “lead the way in innovative solutions by providing surgeons with game-changing operative solutions to better serve them and their patients in hospitals and ambulatory surgical settings.”
“With our expert surgeon design team and leveraging our extensive experience in automated 3D printing and additive manufacturing enabled us to bring a revolutionary new hip replacement system to market.”
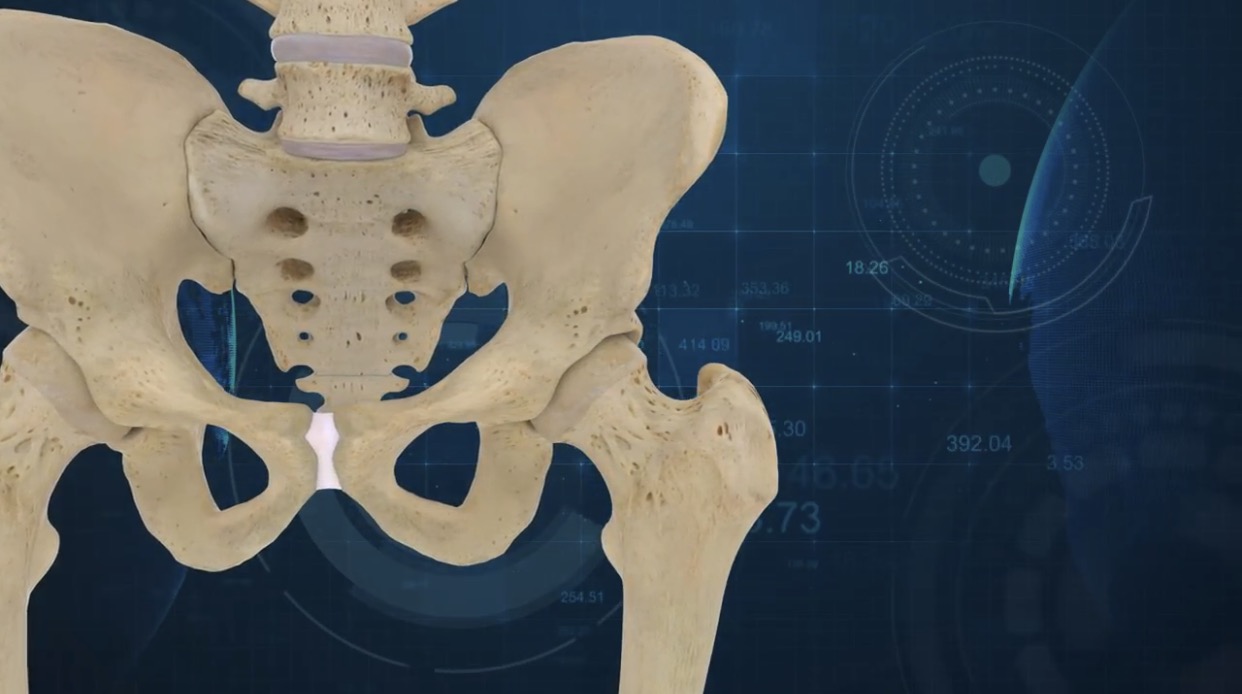
The ConforMIS Hip System
The ConforMIS Hip System uses proprietary advanced imaging and design software, to deliver a suite of FDA-cleared patient conforming knee and hip replacement implants. Following the conversion of a patient’s CT scan into a 3D computer model, the distinctive measurements are transformed into a pre-operative surgical plan that is delivered to the surgeon in advance of the operation.
With patient-specific 3D printed components, the Conformis Hip System is delivered directly to the hospital or surgery center in a single patient-labeled kit, eliminating the need for excess inventory. This includes patient-conforming, single-use, 3D printed cutting guides which reduces the need for reusable instruments required for a standard total hip replacement.
Furthermore, surgeons are able to collaborate with Conformis during the pre-surgical planning phase to design the optimal hip system for each of their patients. Augusti added:
“Our 3D implant design process provides surgeons with interactive input and improves operational efficiencies compared to 2D templating. The design process produces specific individualized pre-navigated cutting guides and implants.”
The first two ConforMIS Hip System surgeries were conducted as part of a limited launch while the complete commercial launch is expected to be announced in 2019.
Stay abreast of the latest news in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
On the lookout for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the ConforMIS Hip System. Image via ConforMIS.
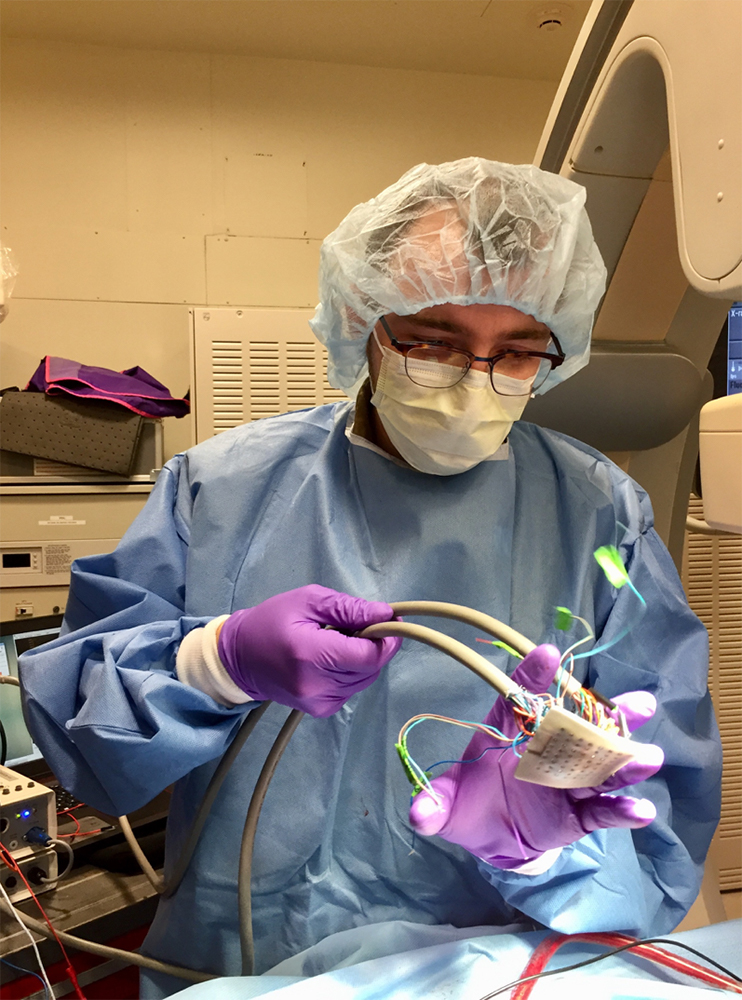
A team of researchers at Stanford University’s School of Medicine, located in California, are developing 3D printed cardiac catheter surgical devices to help those suffering from common heart diseases, such as atrial fibrillation.
Cardiac catheter devices are used by surgeons to map a heart’s electrical activity, ultimately detecting rhythm disturbances in a patient’s heartbeats. However, these devices are often limited to one size, which results in poor connections and missed signals when surveying irregular electrical activity.
Kevin Cyr, a second-year MD student at Stanford Medicine explained that with the use of MRI or CT scans, which records an image file of a patient’s heart, the data can be fed into a 3D printer to “replicate that natural geometry and anatomy specific to that patient.”
Accuracy with every heartbeat
According to Stanford Medicine, atrial fibrillation, a heart condition causing an irregular heart rate, is the most common rhythm disorder, affecting over 6 million Americans every year. This condition disrupts the flow of blood from the heart to the rest of the body, which can lead to blood clots.
Anson Lee, Assistant Professor of Cardiothoracic Surgery at the Stanford University Medical Center began his research into common heart diseases a few years ago and was later joined by Cyr, who became interested in developing new medical technologies.
“Finding and understanding rhythm disturbances in patients has been challenging because of the one-size-fits-all nature of existing medical devices, which use electrodes that contact the surface of the heart to measure electrical activity,” explained Cyr.
Thus, the researchers leveraged additive manufacturing to create customizable cardiac-mapping catheters for each patient, which conform to the unique contours of an individual’s heart.

The 3D printed cardiac-mapping catheters
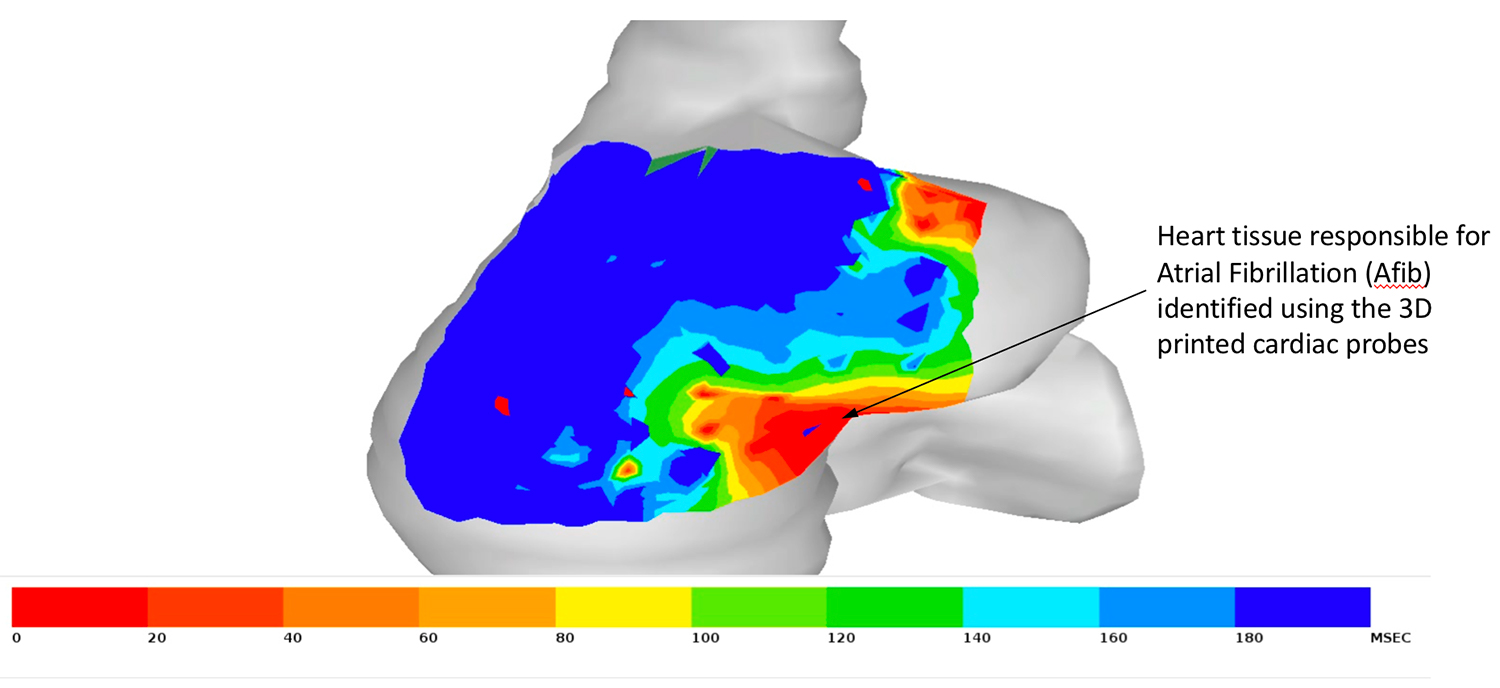
The 3D printed cardiac-mapping catheter device includes a flexible silicone membrane with small apertures in a grid-like formation which holds tiny electrodes. When placed on the surface of the heart’s atrium, the upper chamber in which blood enters the heart, the device surveys the electrical activity over that specific region.
The data is then transmitted to a computer, where it generates a recording of that heartbeat activity. The recordings produce an accurate heatmap of the electrical activity that physicians use to pinpoint the heart regions in need of treatment. Cyr added:
“We can map in perfect detail this rectangular grid of information and not have to worry about missing signals, poor contact or things like that, which otherwise might throw out errors.”
Although this device is only applicable to the external surfaces of the heart, the Stanford research team are investigating whether their device could be used to map the interior surface of the heart – further increasing the measurement accuracy of rhythmic disturbance.
Cyr believes that the devices will likely take another year or two to develop before it is tested on humans.
3D printing and cardiology
Earlier this year, researchers at the Great Ormond Street Hospital (GOSH) in London began developing 3D printed replicas of children’s hearts to be used within the pre-planning surgical process.
Prior to this, the Children’s Heart Research and Outcomes Center (HeRO) developed 3D bioprinted valves, leaflets, and patches to provide a long-lasting solution for children born with heart conditions such as congenital heart disease.
Never miss out on the latest news in 3D printing by subscribing to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Seeking new talent? Or looking for a new position? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows the 3D printed cardiac-mapping catheter devices. Photo via Kevin Cyr.